Chapter 13: Q.13-43P (page 703)
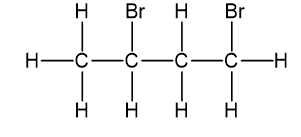
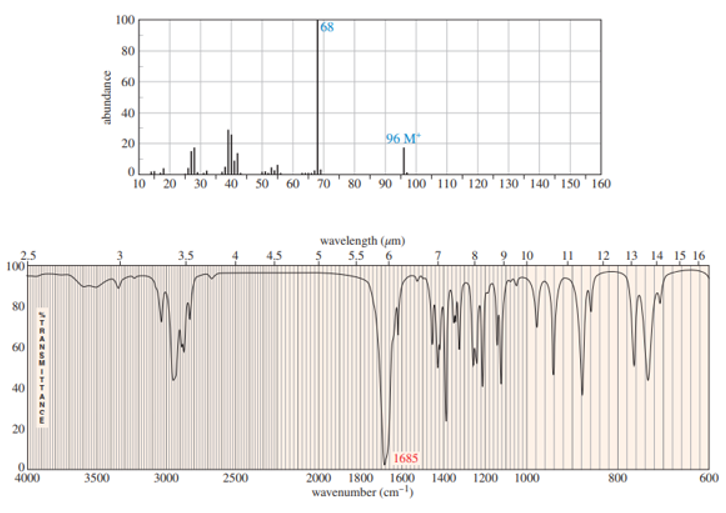
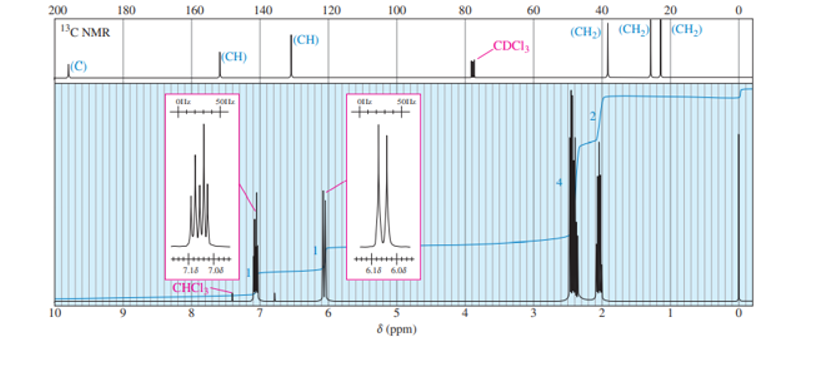
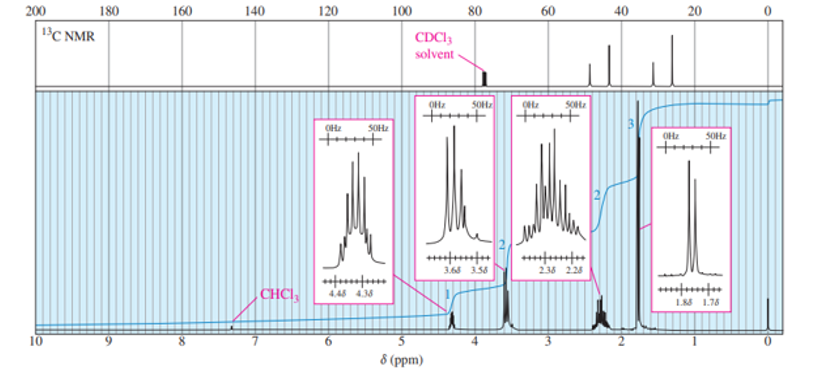
A small pilot was adding bromine across the double bond of but-2-ene to make 2,3-dibromobutane. A controller malfunction allowed the reaction temperature to rise beyond safe limits. A careful distillation of the product showed that several impurities had formed, including the one having the NMR spectra that appear below. Determine its structure and assign the peaks to the protons in your structure.
Short Answer
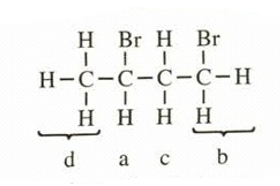
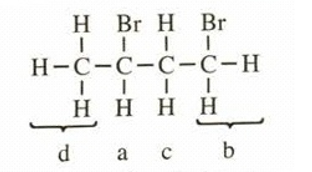
This structure has four different kinds of protons, thus will exhibit four signals in proton NMR.
The values of chemical shifts and splitting of peaks is,
a =4.3 (sextet,
) c =
2.3 (multiplet,
)
b =3.6 (triplet,
) d =
1.7 (doublet,
)