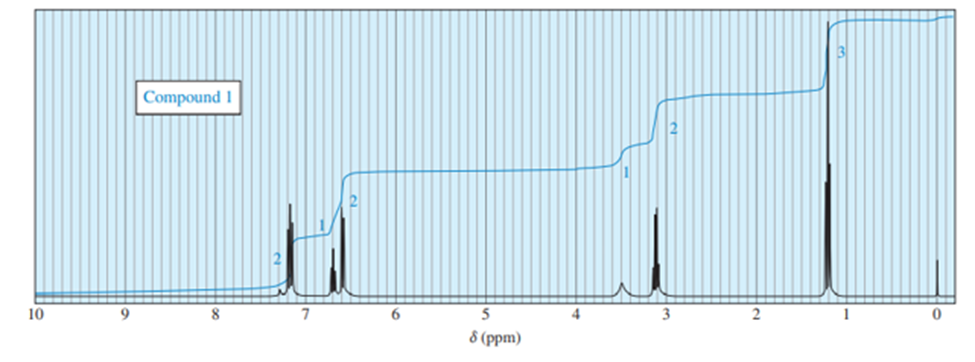
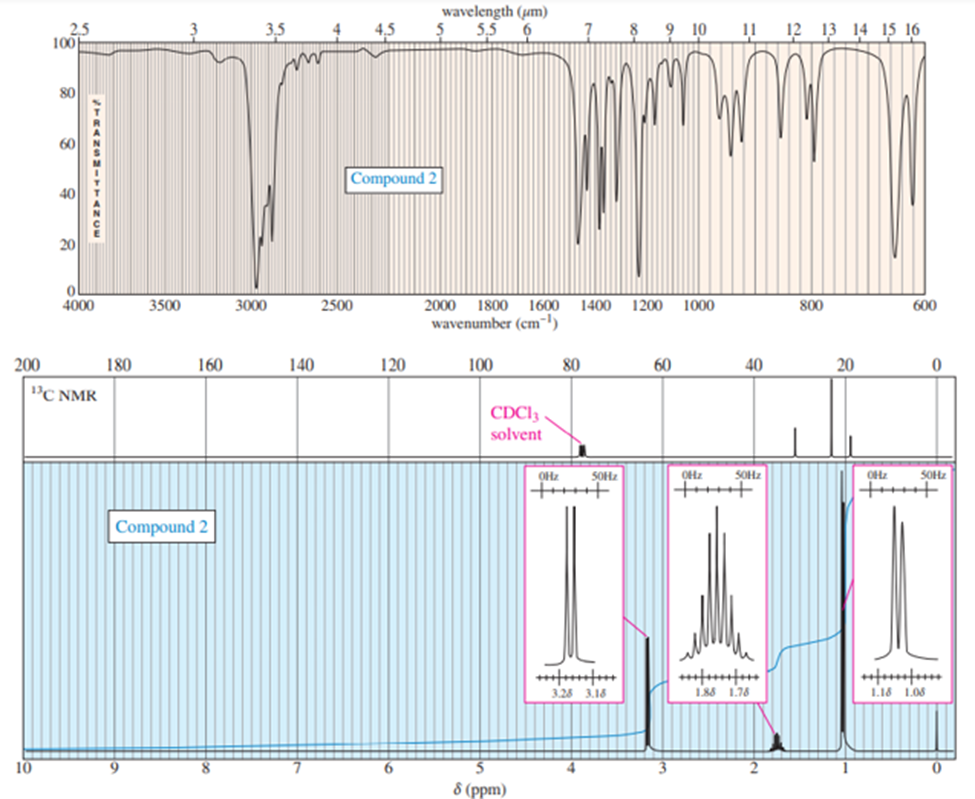
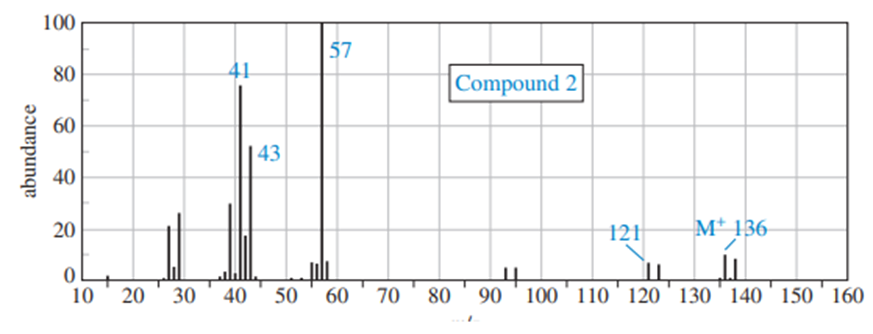
Chapter 13: Q.12P (page 671)
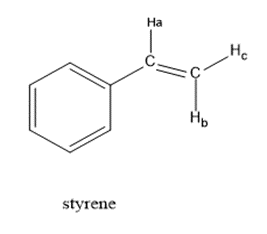
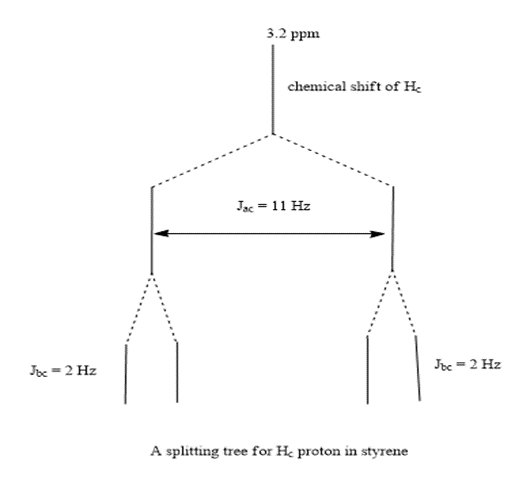
Draw a splitting tree, similar to Figures 13-32 and 13-33, for proton Hc in styrene. What is the chemical shift of proton Hc?
Short Answer
Answer
The splitting tree is:
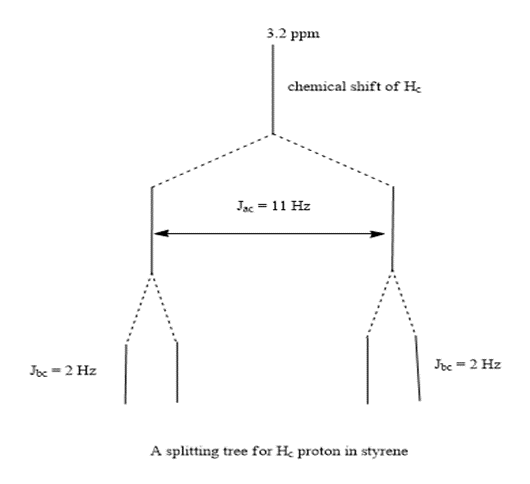
The signal Hc is split ( Jac =11HZ) by coupling with Haand further split( Jbc =2HZ) by coupling with Hb .