Chapter 13: Q36P (page 701)
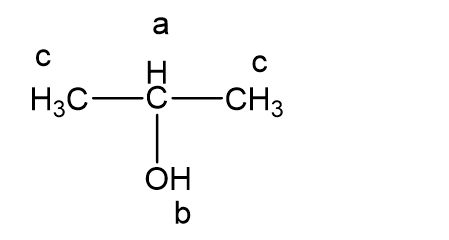
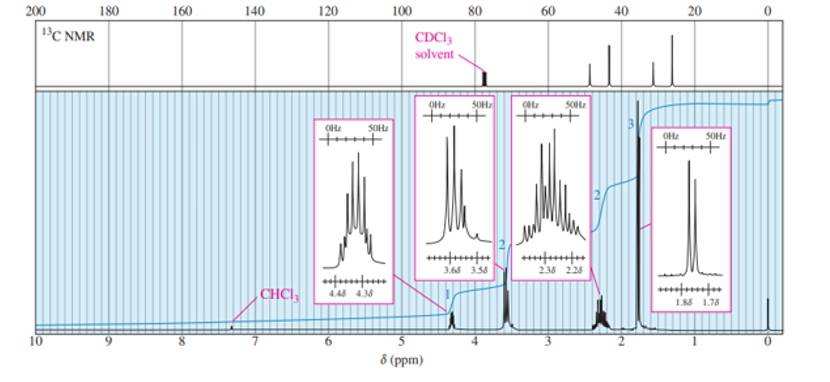
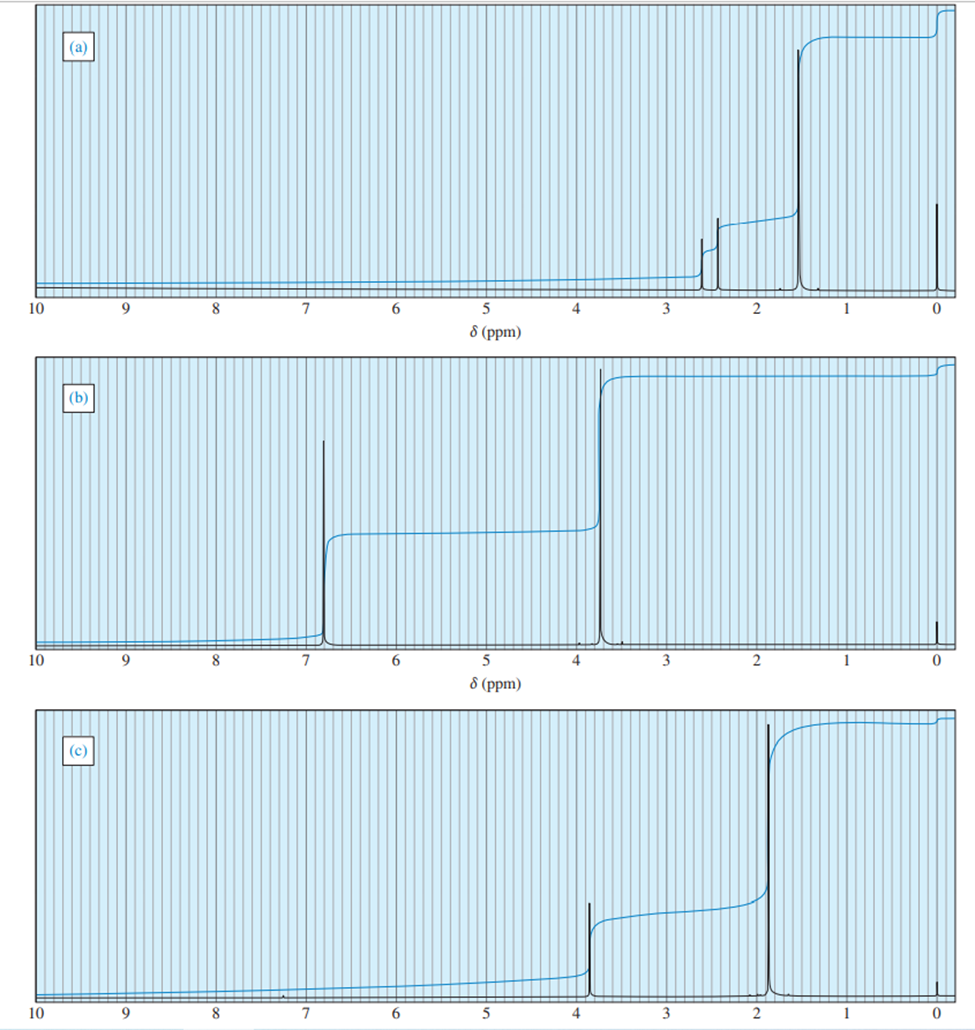
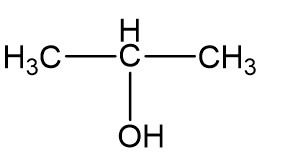
The following proton NMR spectrum is of a compound of molecular formula C3H80.
- Propose a structure for this compound.
- Assign peaks to show which protons give rise to which signals in the spectrum.
Short Answer
a)
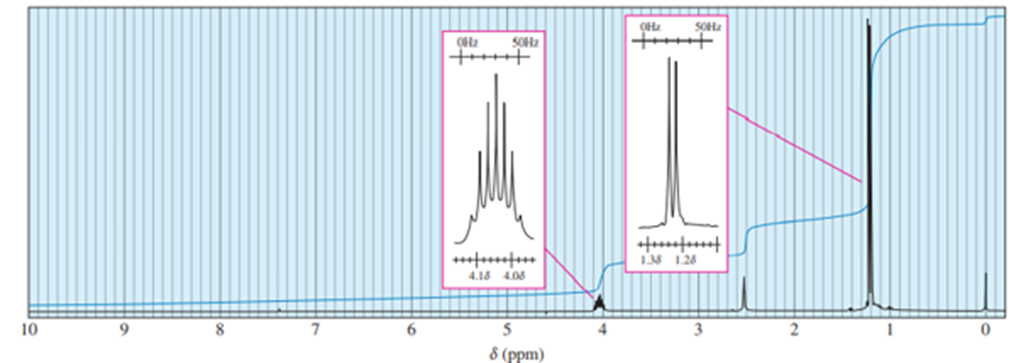
b)
a = δ 4.0 (septet, 1H)
b = δ 2.5 (broad singlet, 1H)
c = δ 1.2 (doublet, 6H)