Chapter 13: Q.13-45P (page 704)
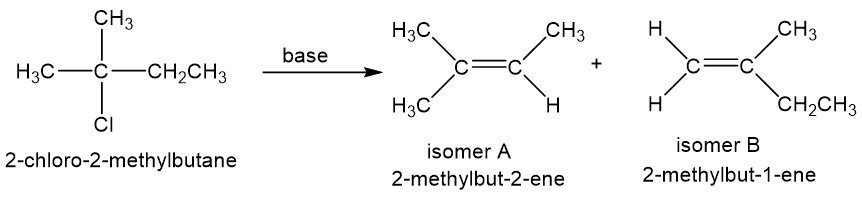
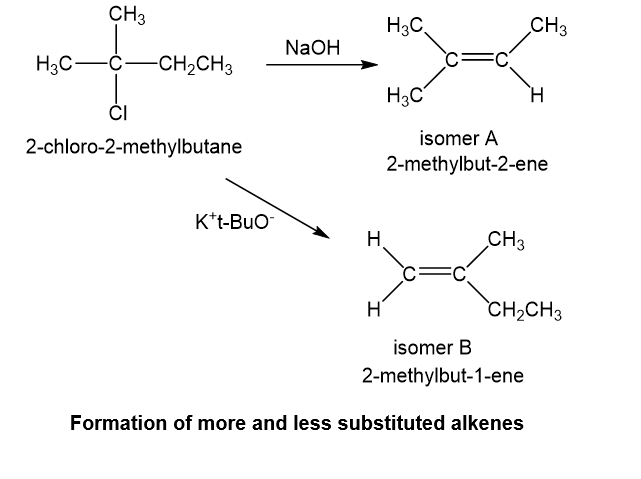
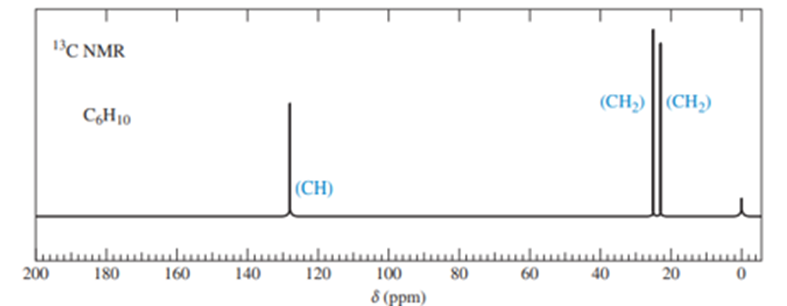
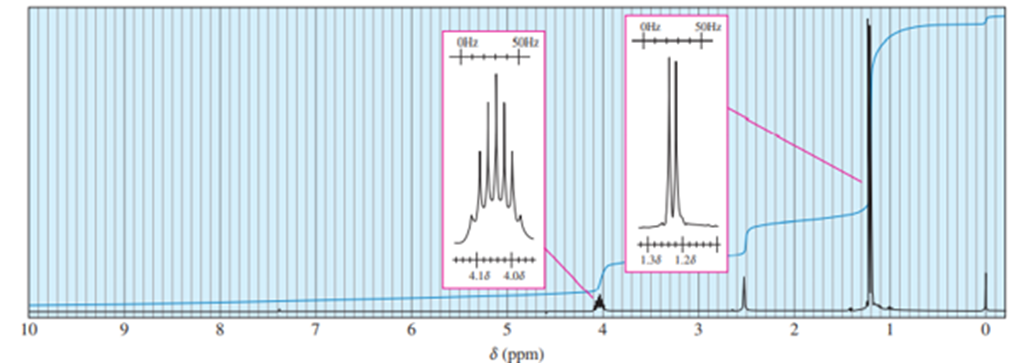
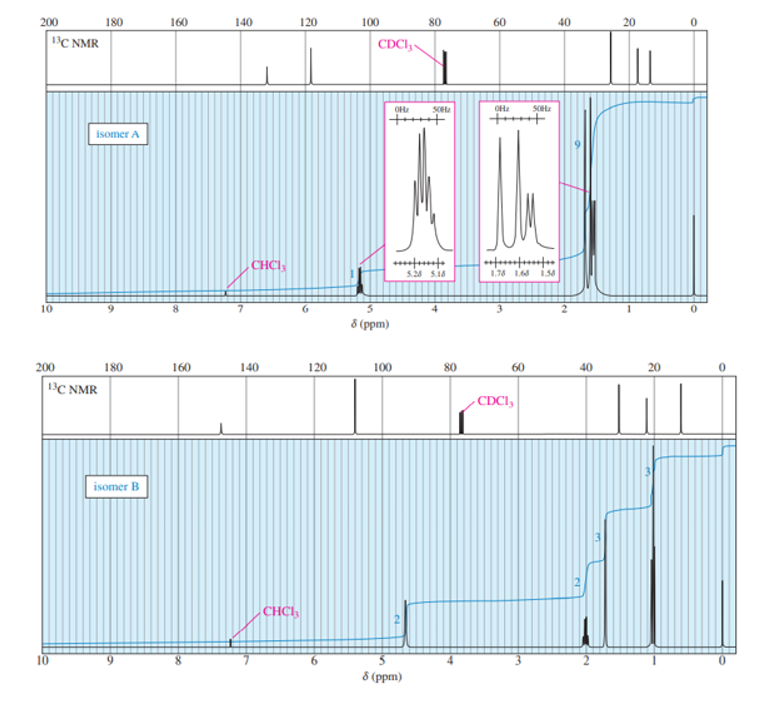
When 2-chloro-2-methylbutane is treated with a variety of strong bases, the products always seem to contain two isomers (A and B) of formula. When sodium hydroxide is used as base, isomer A predominates. When potassium tert-butoxide is used as the base, isomer B predominates. The
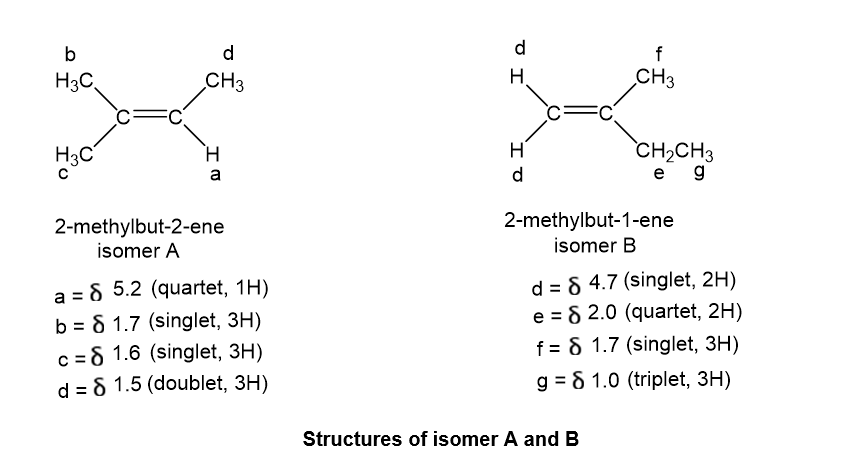
and
NMR spectra of A and B are given below.
- Determine the structures of isomers A and B.
- Explain why A is the major product when using sodium hydroxide as the base and why B is the major product when using potassium tert-butoxide as the base.
Short Answer
a.
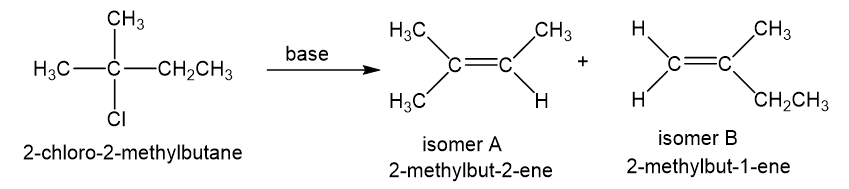
b. With sodium hydroxide as the base, the more highly substituted alkene, that is isomer A would be expected to predominate as per Zaitsev rule. With potassium tert-butoxide as the bulky base, the less substituted alkene, that is, isomer B would predominate, that is the Hofmann product.