Chapter 13: Q.13-55 (page 707)
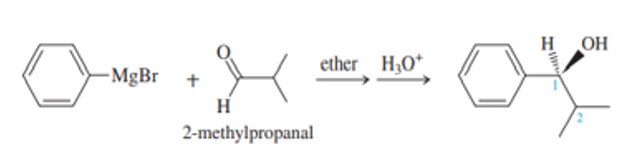
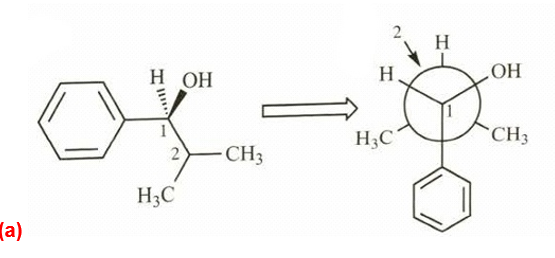
Phenyl Grignard reagent adds to 2-methylpropanal to give the secondary alcohol shown. The proton NMR of 2-methylpropanal shows the two methyl groups as equivalent (one doublet at1.1), yet the product alcohol, a racemic mixture, shows two different
doublets, one at
0.75 and one around
1.0.
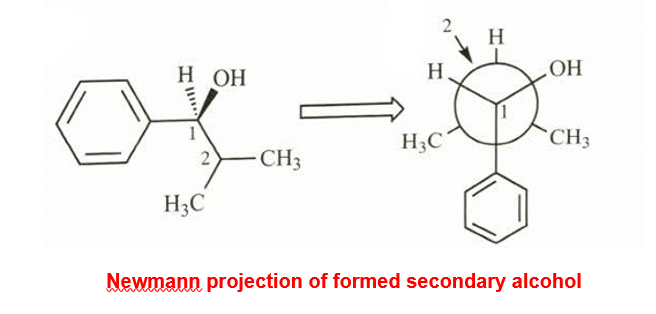
- Draw a Newmann projection of the product along the C1-C2 axis.
- Explain why the two methyl groups have different NMR chemical shifts. What is the term applied to protons such as these?
Short Answer
(B)The two methyl groups are diastereotopic in nature and have different values of chemical shifts