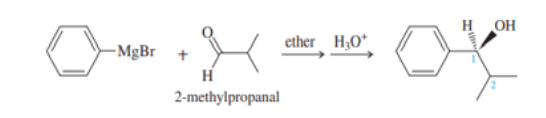
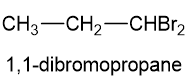Chapter 13: Q.13-50P (page 706)
(a) Draw all five isomers of formula(including stereoisomers).
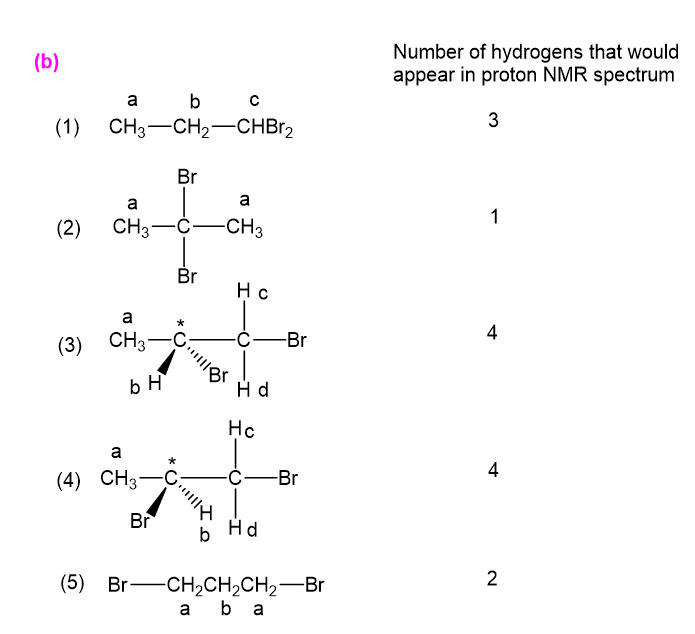
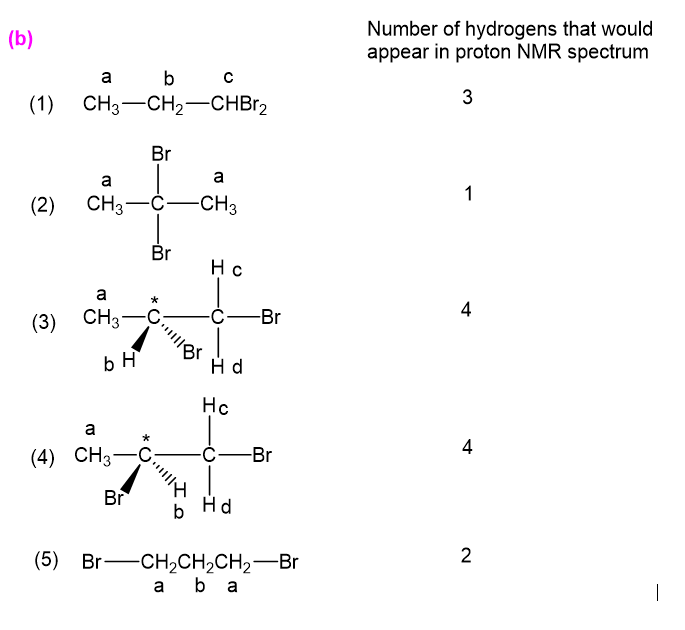
(b) For each structure, show how many types of would appear in the proton NMR spectrum.
(c) For each structure, show how many types ofwould appear in theNMR spectrum.
(d) If an unknown compound of formulashows three types of and three types of ,can you determine its structure from this information?
Short Answer
(a)There are five isomers of formula , including stereoisomers, these are the following:
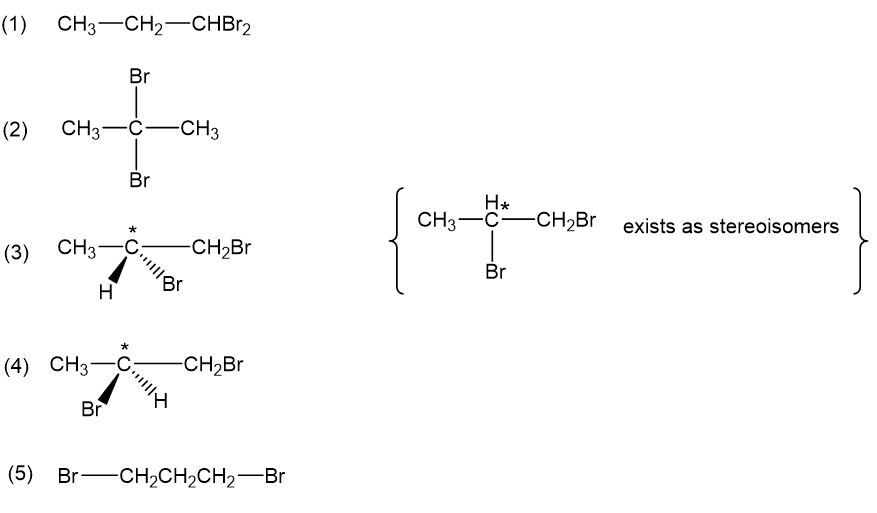
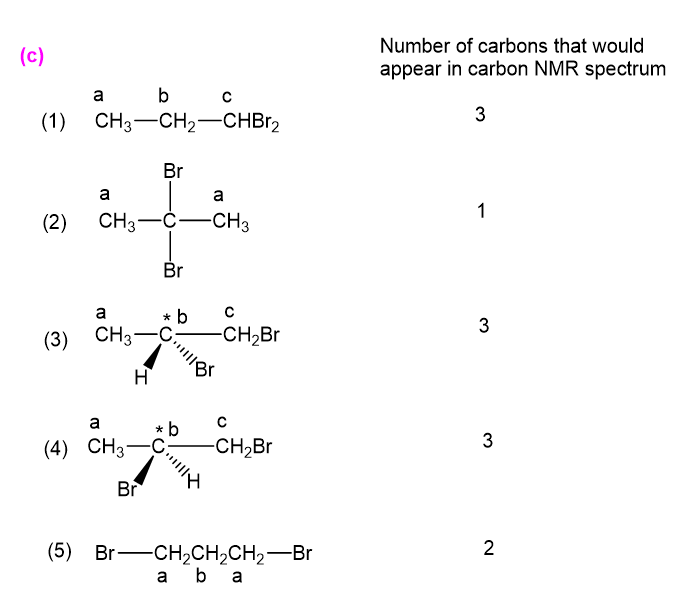
(d)If an unknown compound of formula shows three types of and three types of, then the proposed structure will be 1,1-dibromopropane.
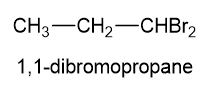
 , these are:
, these are: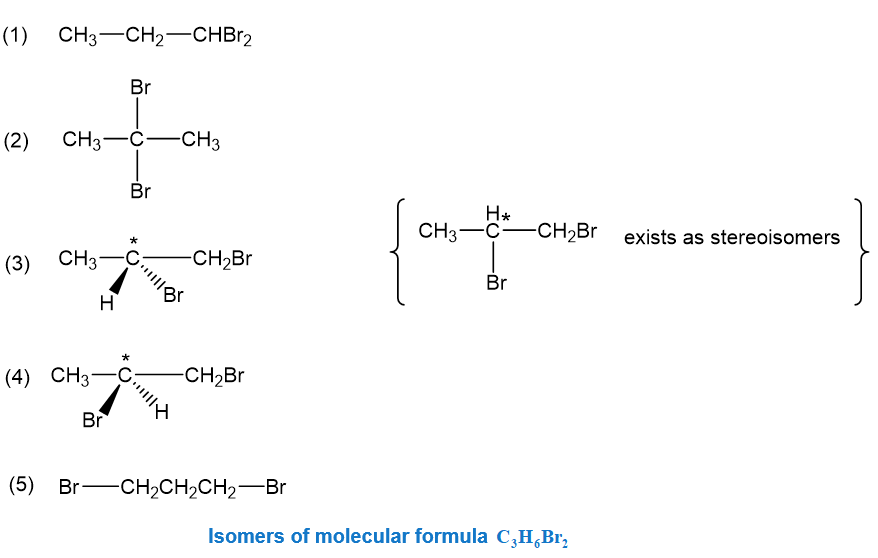
 have been marked a,b,c,d in the following structures:
have been marked a,b,c,d in the following structures: