Chapter 18: Q57P (page 971)
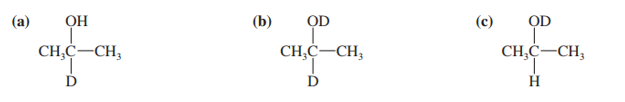
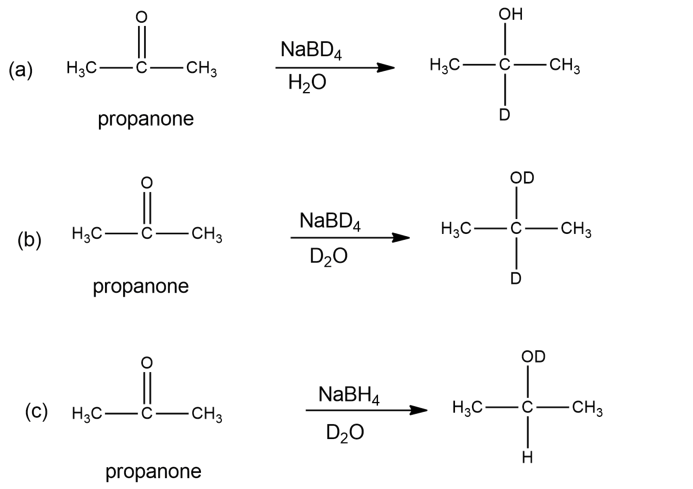
Both NaBH4 and NaBD4 are commercially available, and D2O is common and inexpensive. Show how you would synthesize the following labelled compounds, starting with propanone.
Short Answer
Learning Materials
Features
Discover
Chapter 18: Q57P (page 971)
Both NaBH4 and NaBD4 are commercially available, and D2O is common and inexpensive. Show how you would synthesize the following labelled compounds, starting with propanone.


All the tools & learning materials you need for study success - in one app.
Get started for free
Question: The family of macrolide antibiotics all have large rings (macrocycle) in which an ester is what makes the ring; a cyclic ester is termed as a lactone. One example is amphotericin B, used as an anti-fungal treatment of last resort because of its liver and heart toxicity. Professor Martin Burke of the University of Illnois has been making analogs to retain the antifungal properties but without the toxicity, including this structure published in 2015. (Nature Chemical Biology, (2015) doi: 10.1038/nchembio.1821). The carboxylate of amphotericin B has been replaced with the urea group (shown in red).
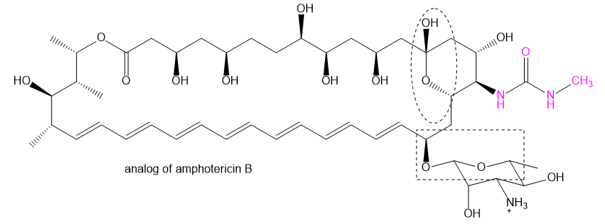
(a) Where is the lactone group that forms the ring?
(b) Two groups are circled. What type of functional group are they? Explain.
Give the structures of the carbonyl compound and the amine used to form the following imines.
(a)
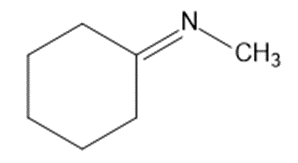
(b)
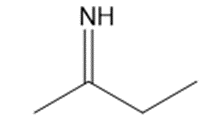
(c)
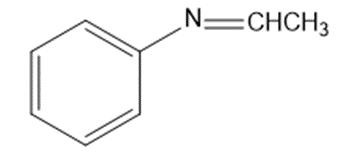
(d)
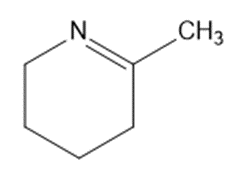
(e)
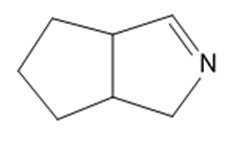
(f)
Why were no products from McLafferty rearrangement observed in the spectrum of butan-2-one?
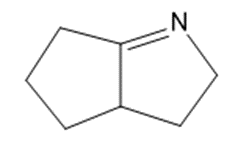
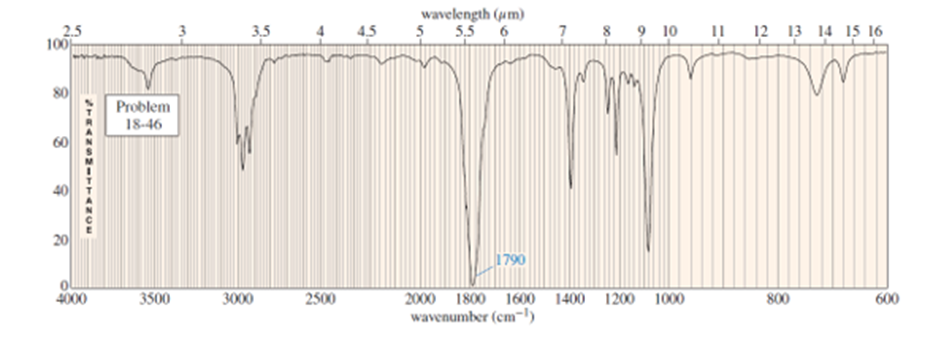
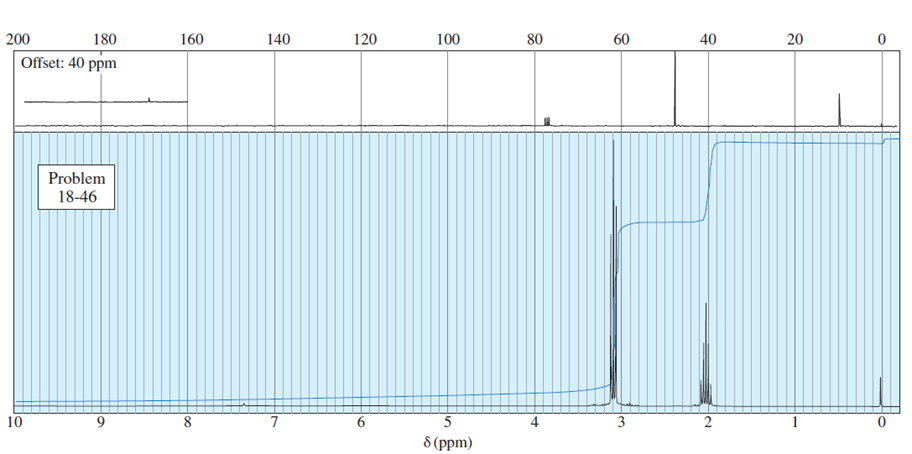
Question. An unknown compound gives a molecular ion of m/z 70 in the mass spectrum. It reacts with semicarbazide hydrochloride to give a crystalline derivative, but it gives a negative Tollens test. The NMR and IR spectra follow. Propose a structure for this compound, and give peak assignments to account for the absorptions in spectra. Explain why the signal atin the IR spectrum appears at an unusual frequency.
Show how you would accomplish the following syntheses.
(a).
(b).
(c).
(d).
What do you think about this solution?
We value your feedback to improve our textbook solutions.