(a) Cyclopentanecarbaldehyde on treatment with phenylmagnesium bromide in presence of an acidic catalyst H+ givesthe product as cyclopentyl(phenyl)methanol.
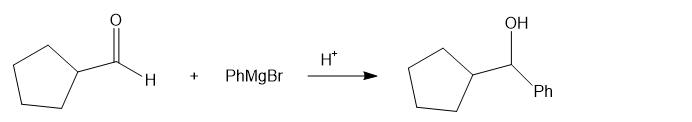
reaction a
(b) Cyclopentanecarbaldehyde on treatment with tollens reagent to givethe products as silver mirror, ammonia and cyclopentanecarboxylic acid.

reaction b
(c) Cyclopentanecarbaldehyde on treatment with hydrazinecarboxamide in presence of an weak acid givesthe product as 2-(cyclopentylmethyl)hydrazinecarboxamide.
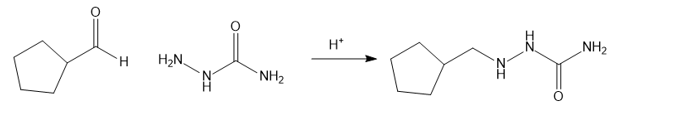
reaction c
(d) Cyclopentanecarbaldehyde on treatment with excess ethanol and acid give sthe product as (diethoxymethyl)cyclopentane.
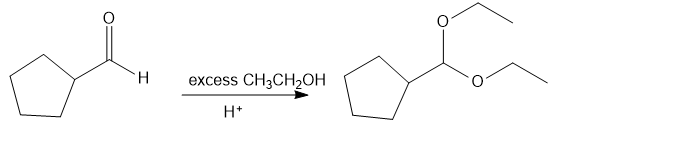
reaction d
(e) Cyclopentanecarbaldehyde on treatment with propane-1,3-diol in presence of an acidic catalyst H+ givesthe product as 2-cyclopentyl-1,3-dioxane.
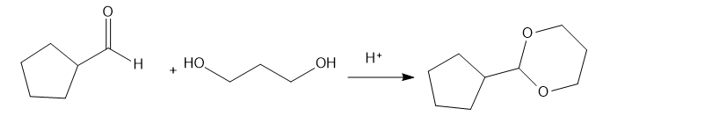
reaction e
(f) Cyclopentanecarbaldehyde on treatment with amalgamated zinc (zinc treated with mercury) and hydrochloric aacid gives the product as methylcyclopentane
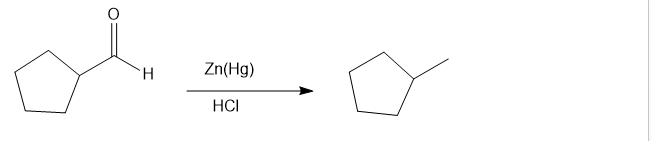
reaction f



















