Chapter 18: Q33P (page 957)
Show how Wittig reactions might be used to synthesize the following compounds. In each case, start with an alkyl halide and a ketone or an aldehyde.
(a)
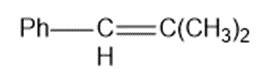
(b)
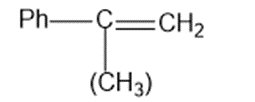
(c)
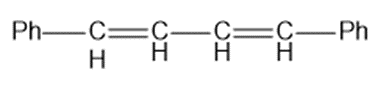
(d)
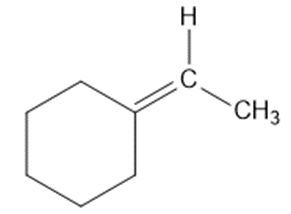
Short Answer
(a)
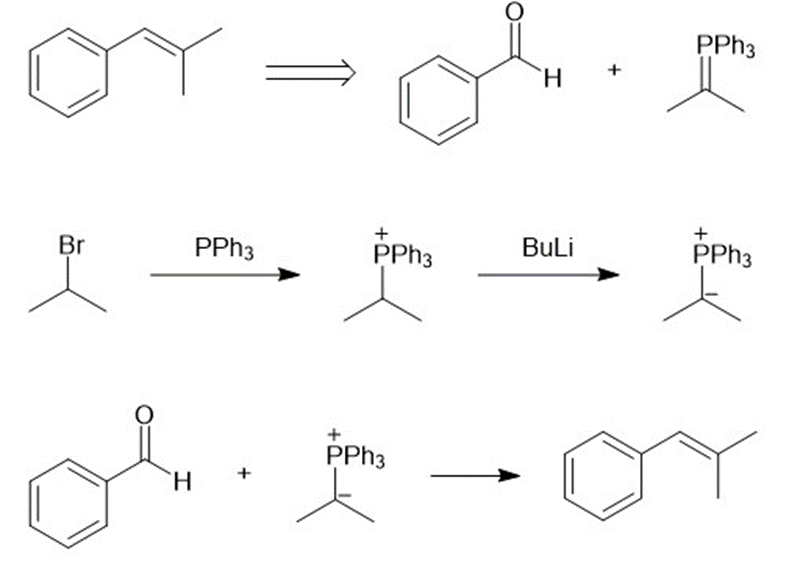
(b)
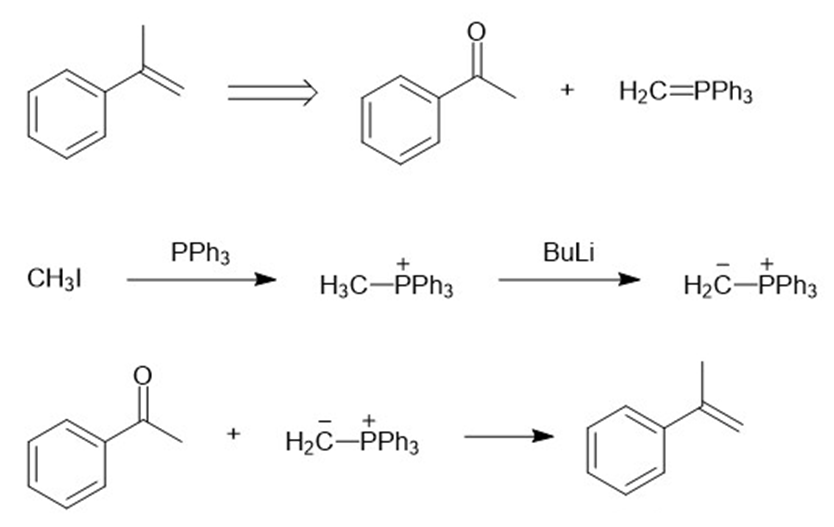
(c)
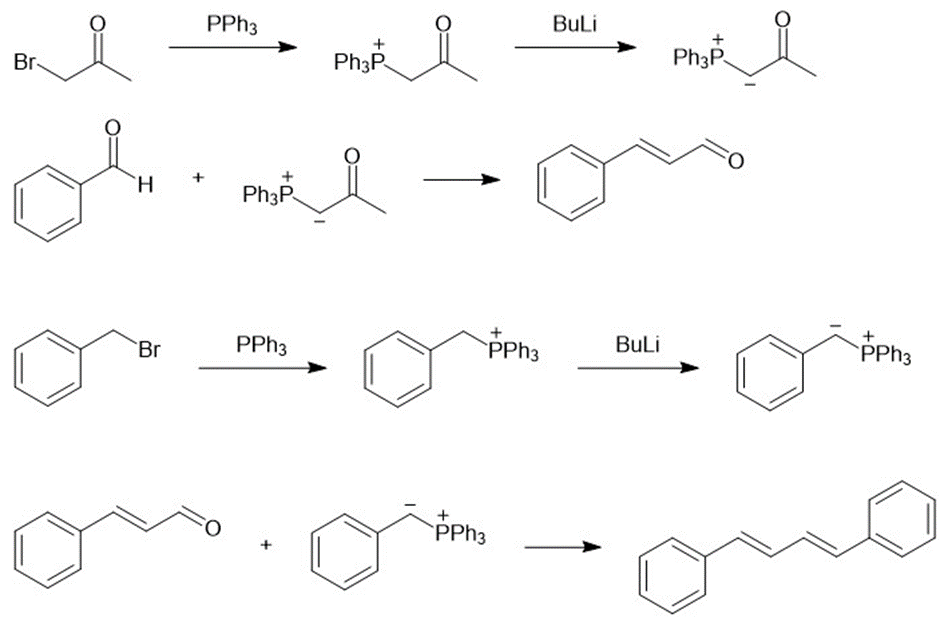
(d)
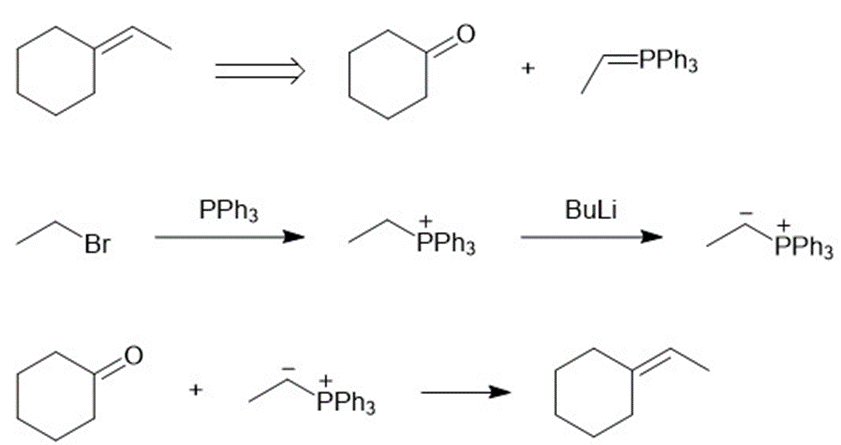
Learning Materials
Features
Discover
Chapter 18: Q33P (page 957)
Show how Wittig reactions might be used to synthesize the following compounds. In each case, start with an alkyl halide and a ketone or an aldehyde.
(a)
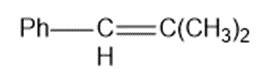
(b)

(c)

(d)

(a)

(b)

(c)

(d)

All the tools & learning materials you need for study success - in one app.
Get started for free
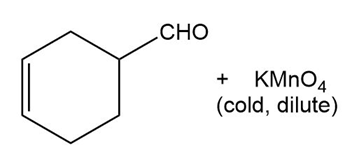
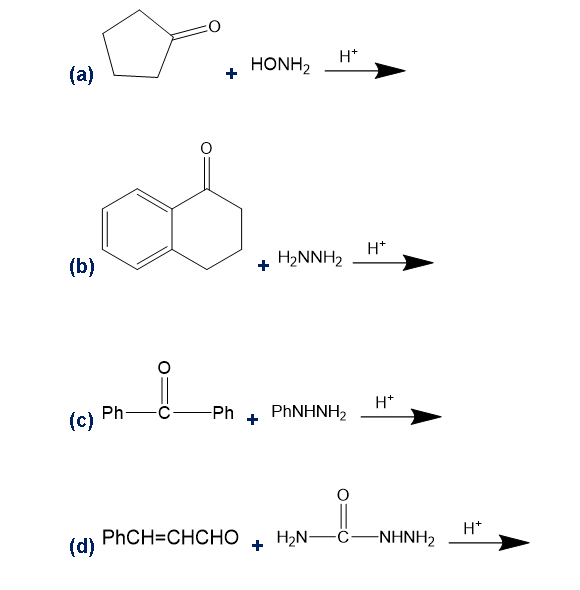
Predict the major products of the following reactions.
(a) (b)

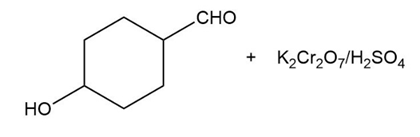
(c) (d)

Question: The family of macrolide antibiotics all have large rings (macrocycle) in which an ester is what makes the ring; a cyclic ester is termed as a lactone. One example is amphotericin B, used as an anti-fungal treatment of last resort because of its liver and heart toxicity. Professor Martin Burke of the University of Illnois has been making analogs to retain the antifungal properties but without the toxicity, including this structure published in 2015. (Nature Chemical Biology, (2015) doi: 10.1038/nchembio.1821). The carboxylate of amphotericin B has been replaced with the urea group (shown in red).
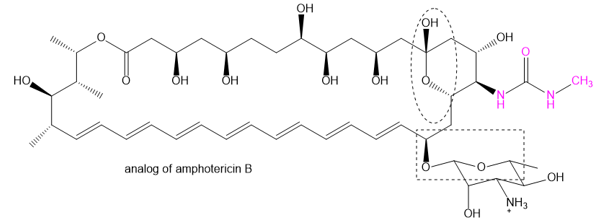
(a) Where is the lactone group that forms the ring?
(b) Two groups are circled. What type of functional group are they? Explain.
Predict the products of the following reactions.
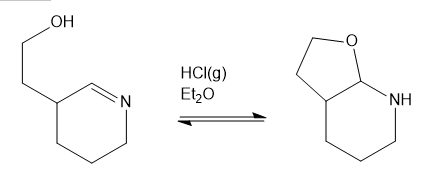
Question: Show a complete mechanism for this equilibrium established in diethyl ether with HCI gas as catalyst.
Sketch the expected proton NMR spectra of 3,3- dimethylbutanal.
What do you think about this solution?
We value your feedback to improve our textbook solutions.