Chapter 18: Q69P (page 912)
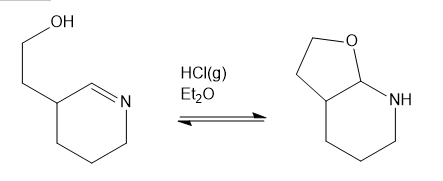
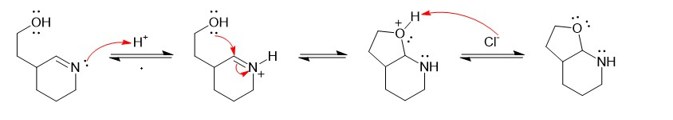
Question: Show a complete mechanism for this equilibrium established in diethyl ether with HCI gas as catalyst.
Short Answer
Learning Materials
Features
Discover
Chapter 18: Q69P (page 912)
Question: Show a complete mechanism for this equilibrium established in diethyl ether with HCI gas as catalyst.


All the tools & learning materials you need for study success - in one app.
Get started for free
Predict the products formed when cyclopentanecarbaldehyde reacts with the following reagents.
(a) PhMgBr, then H3O+
(b) Tollens reagent
(c) semicarbazide and weak acid
(d) excess ethanol and acid
(e) propane-1-3-diol, H+
(f) zinc amalgam and dilute hydrochloric acid
Show what alcohols and carbonyl compounds give the following derivatives.
(a)
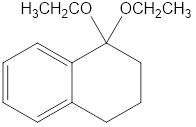
(b)
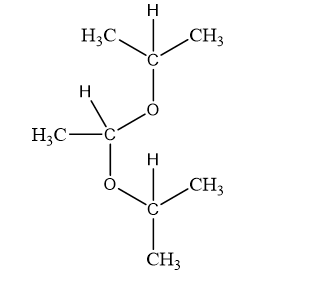
(c)
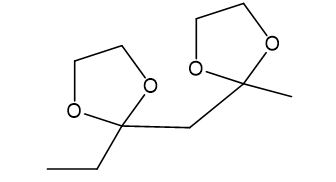
(d)
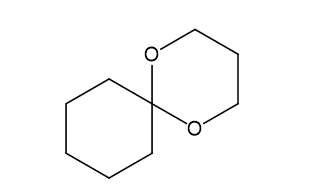
(e)
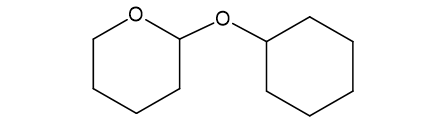
(f)
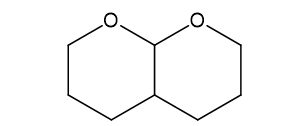
Predict the products formed when cyclopentanone reacts with the following reagents.
(a) CH3NH2 , H+
(b) excess CH3OH , H+
(c) hydroxylamine and weak acid
(d) ethylene glycol and p-toluenesulfuric acid
(e) phenylhydrazine and weak acid
(f) PhMgBr and then mild H3O+
(g) Tollens reagent
(h) sodium acetlylide, then H3O+
(i) hydrazine, then hot. fused KOH
(j) Ph3P=CH2
(k) sodium cyanide
(l) acidic hydrolysis of the product from (k)
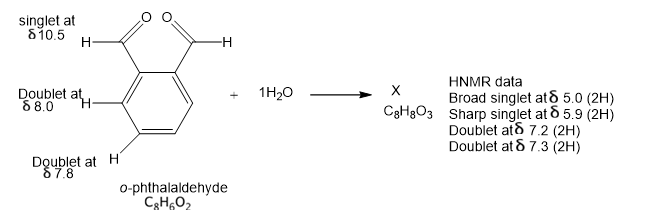
In the absence of water, ophthalaldehyde has the structure shown. Its strong IR absorption is at 1687 cm-1; the proton NMR data are shown by the structure. In the presence of water, a new compound is formed that has a strong IR absorption around 3400 cm-1 and no absortion in the C=0 region. propose a structure of X consistent with this information and suggest how X consistent with this information and suggest how X was formed.
Sodium triacetoxyborohydride, , is a mild reducing agent that reduces aldehydes more quickly than ketones. It can be used to reduce aldehydes in the presence of ketones, such as in the following reaction:
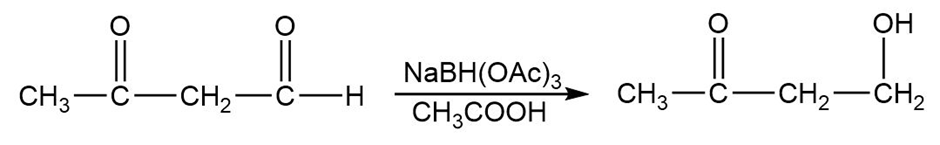
(a) Draw a complete Lewis structure for sodium triacetoxyborohydride.
(b) Propose a mechanism for the reduction of an aldehyde by sodium triacetoxyborohydride.
What do you think about this solution?
We value your feedback to improve our textbook solutions.