Chapter 18: Q6P (page 929)
Show how you would synthesize each compound from starting materials containing no more than six carbon atoms.
(a)
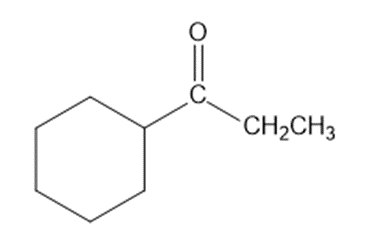
(b)
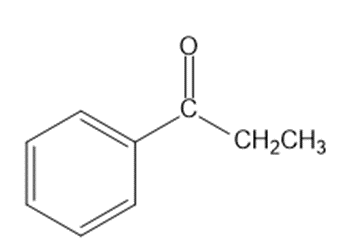
(c)
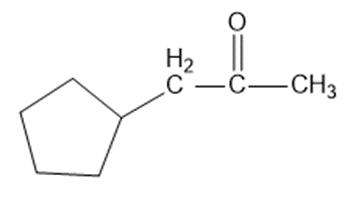
Short Answer
(a)
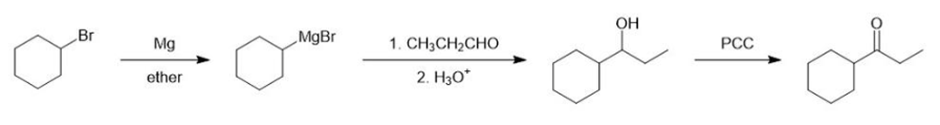
(b)
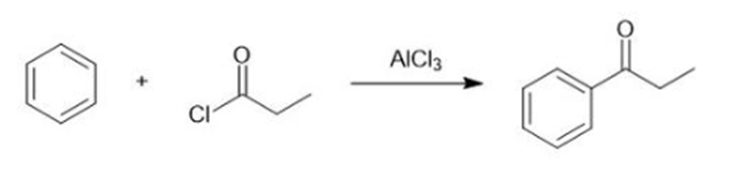
(c)
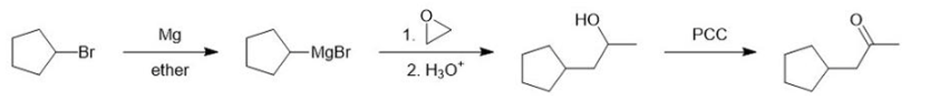
Learning Materials
Features
Discover
Chapter 18: Q6P (page 929)
Show how you would synthesize each compound from starting materials containing no more than six carbon atoms.
(a)

(b)

(c)

(a)

(b)

(c)

All the tools & learning materials you need for study success - in one app.
Get started for free
The Wittig reaction is useful for placing double bonds in less stable positions. For example, the following transformation is easily accomplished using a Wittig reaction
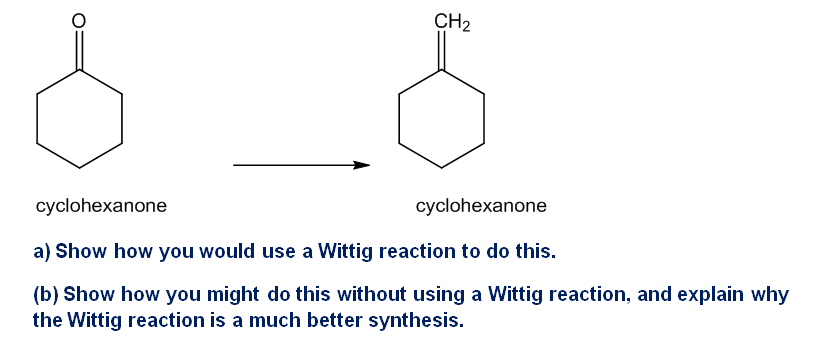
Depending on the reaction conditions, two different imines of formula might be formed by the reaction of benzaldehyde with methylamine. Explain, and give the structures of two imines.
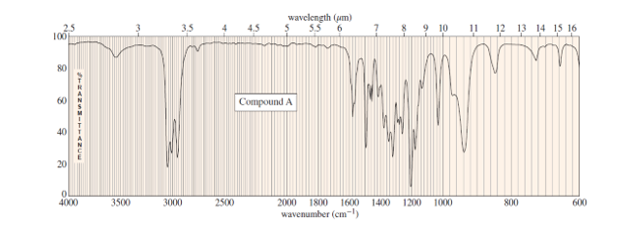
Question. The mass spectrum of unknown compound A shows a molecular ion at m/z 116 and prominent peaks at m/z 87 and m/z 101. Its UV spectrum shows no maximum above 200 nm. The IR and NMR spectra of A follow. When A is washed with dilute aqueous acid, extracted into dichloromethane , and the solvent evaporated, it gives a product B. B shows a strong carbonyl signal at 1715 cm-1in the IR spectrum and a weak maximum at 274nm(E =16) in the UV spectrum. The mass spectrum of B shows a molecular ion of m/z 72. Determine the structures of A and B, and show the fragmentation to account for the peaks at m/z 87 and 101.
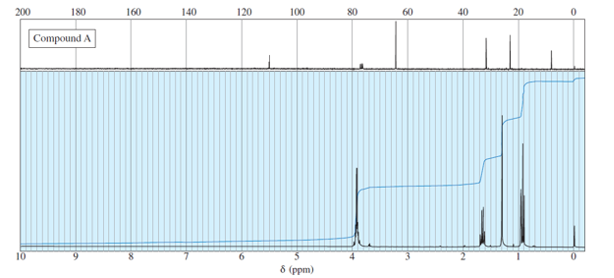
Question. An unknown compound gives a molecular ion of m/z 70 in the mass spectrum. It reacts with semicarbazide hydrochloride to give a crystalline derivative, but it gives a negative Tollens test. The NMR and IR spectra follow. Propose a structure for this compound, and give peak assignments to account for the absorptions in spectra. Explain why the signal atin the IR spectrum appears at an unusual frequency.
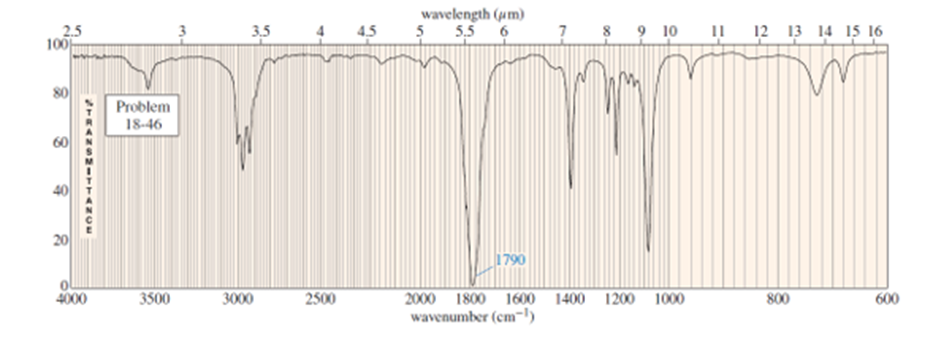
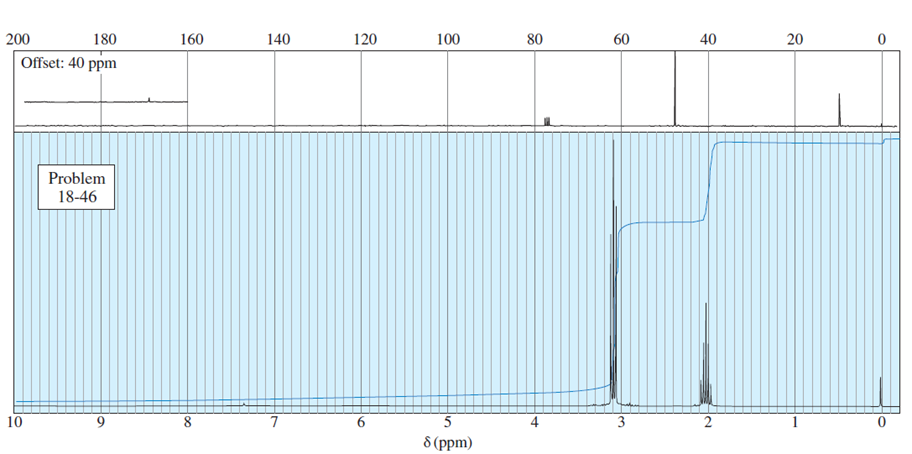
What do you think about this solution?
We value your feedback to improve our textbook solutions.