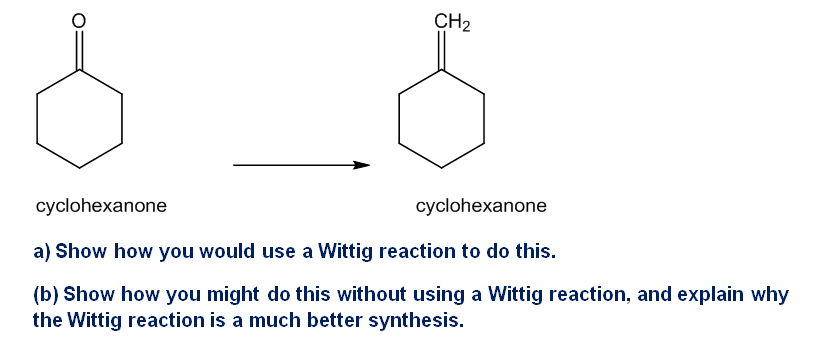
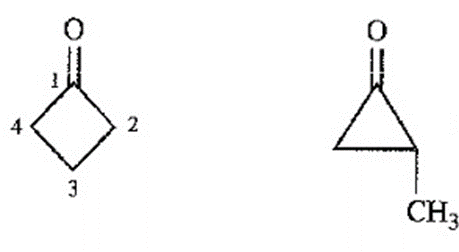Chapter 18: Q.46P (page 970)
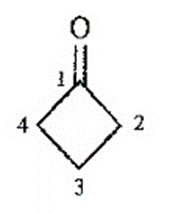
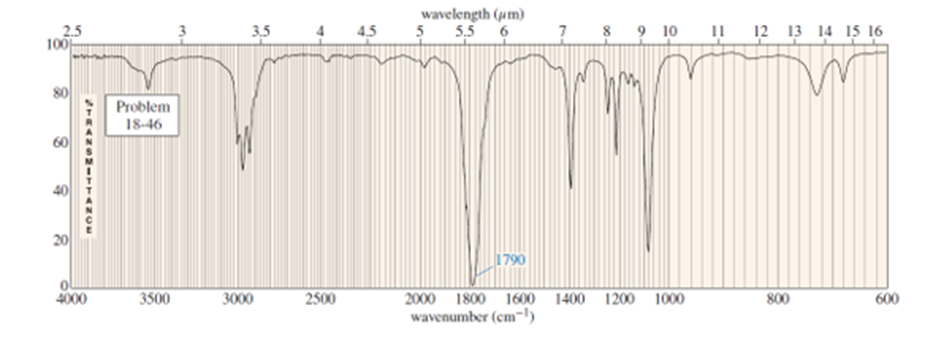
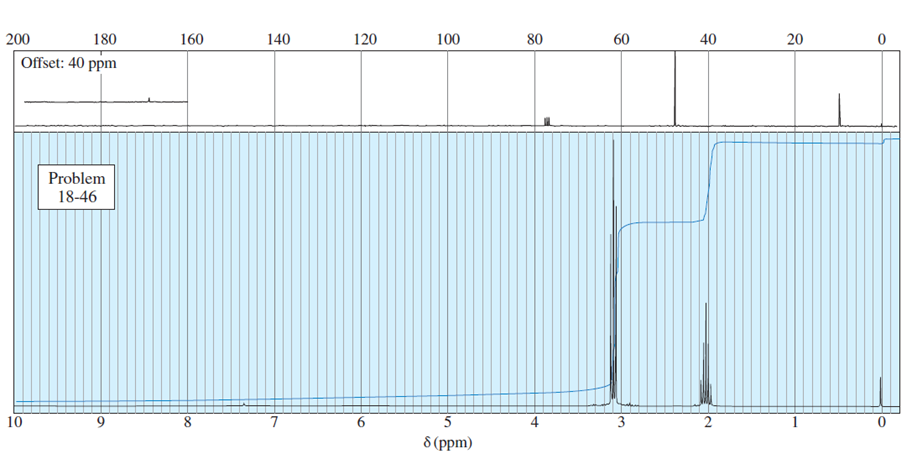
Question. An unknown compound gives a molecular ion of m/z 70 in the mass spectrum. It reacts with semicarbazide hydrochloride to give a crystalline derivative, but it gives a negative Tollens test. The NMR and IR spectra follow. Propose a structure for this compound, and give peak assignments to account for the absorptions in spectra. Explain why the signal atin the IR spectrum appears at an unusual frequency.
Short Answer
Answer
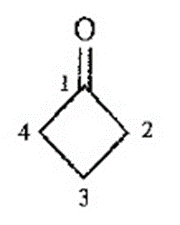
Cyclobutanone