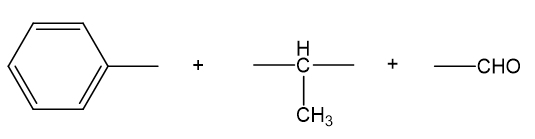
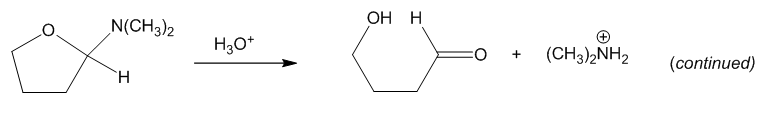
Chapter 18: Q2P (page 920)
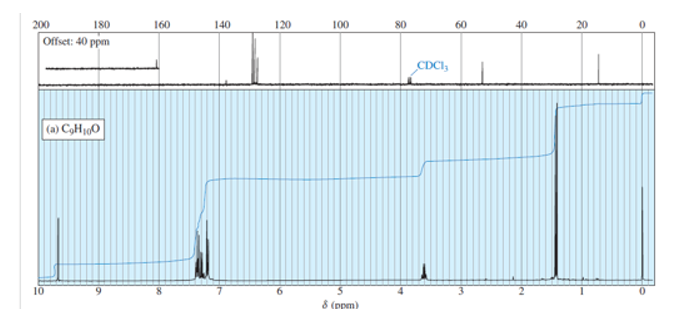
NMR spectra for two compounds are given here, together with the molecular formulas. Each compound is a ketone or an aldehyde. In each case, show what characteristics of the spectrum imply the presence of a ketone or an aldehyde, and propose a structure for the compound.
Short Answer
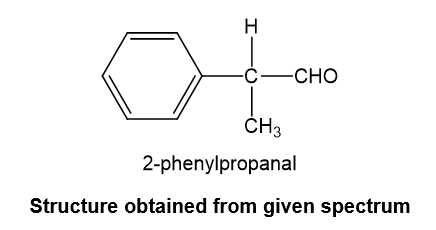
Structure obtained from the given spectrum is,
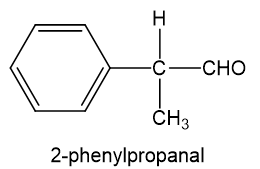
has five elements of unsaturation. doublet with very small coupling constant is observed at chemical shift value 9.7 indicates aldehydic hydrogen next to data-custom-editor="chemistry" . data-custom-editor="chemistry" multiple peaks at chemical shift value 7.2-7.4 indicates a monosubstituted benzene. multiplet at chemical shift value 3.6 and doublet at chemical shift value 1.4 indicates . Combining all the fragments together, we get the proposed structure to be 2-phenylpropanal.