Chapter 18: Q15P (page 942)
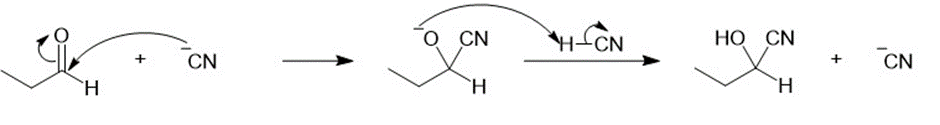
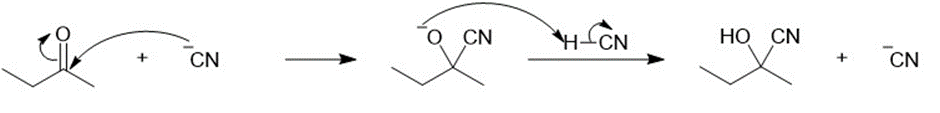
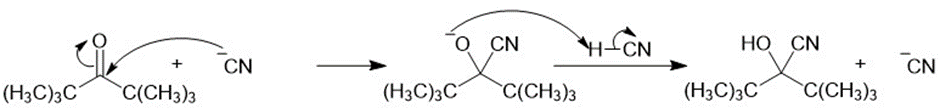
Propose a mechanism for each cyanohydrin synthesis just shown.
Short Answer
Learning Materials
Features
Discover
Chapter 18: Q15P (page 942)
Propose a mechanism for each cyanohydrin synthesis just shown.



All the tools & learning materials you need for study success - in one app.
Get started for free
Hydration of alkynes (via oxymercuration) gives good yields of single compounds only with symmetrical or asymmetrical alkynes. Show what products would be from hydration fo each compound.
(a) hex-3-yne
(b) hex-2-yne
(c) hex-1-yne
(d) cyclodecyne
(e) 3-methylcyclodecyne
For each compound,
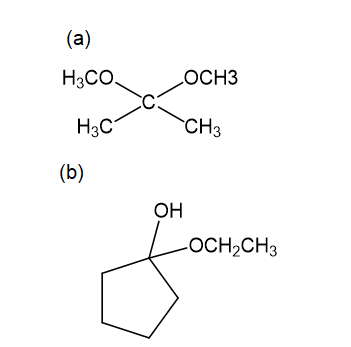
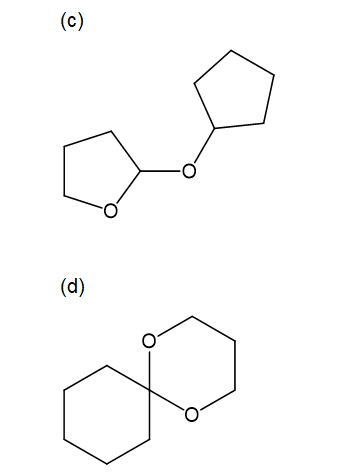
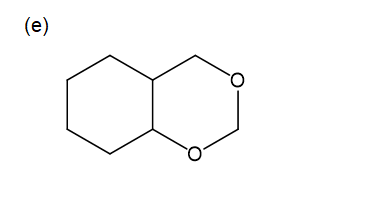
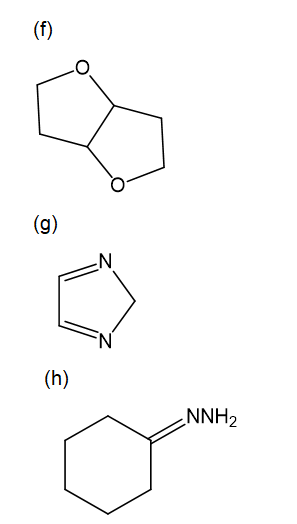
Propose mechanisms for
(a) the acid-catalyzed hydration of chloral to form chloral hydrate.
(b) the base-catalyzed hydration of acetone to form acetone hydrate
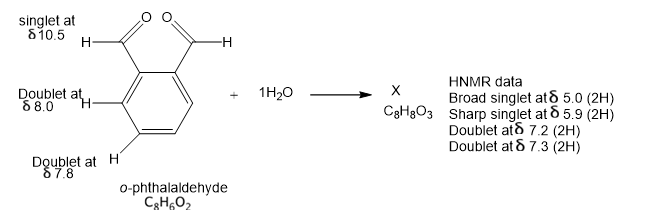
In the absence of water, ophthalaldehyde has the structure shown. Its strong IR absorption is at 1687 cm-1; the proton NMR data are shown by the structure. In the presence of water, a new compound is formed that has a strong IR absorption around 3400 cm-1 and no absortion in the C=0 region. propose a structure of X consistent with this information and suggest how X consistent with this information and suggest how X was formed.
Propose a mechanism for the acid-catalyzed reaction of benzaldehyde with methanol to give benzaldehyde dimethyl acetal.
What do you think about this solution?
We value your feedback to improve our textbook solutions.