Question: The family of macrolide antibiotics all have large rings (macrocycle) in which an ester is what makes the ring; a cyclic ester is termed as a lactone. One example is amphotericin B, used as an anti-fungal treatment of last resort because of its liver and heart toxicity. Professor Martin Burke of the University of Illnois has been making analogs to retain the antifungal properties but without the toxicity, including this structure published in 2015. (Nature Chemical Biology, (2015) doi: 10.1038/nchembio.1821). The carboxylate of amphotericin B has been replaced with the urea group (shown in red).
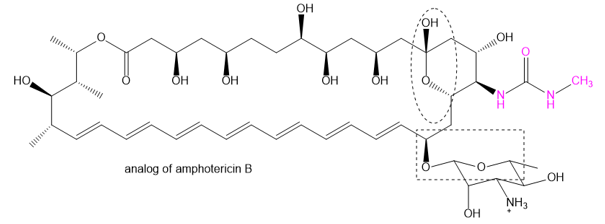
(a) Where is the lactone group that forms the ring?
(b) Two groups are circled. What type of functional group are they? Explain.

















