Chapter 18: Q4P (page 923)
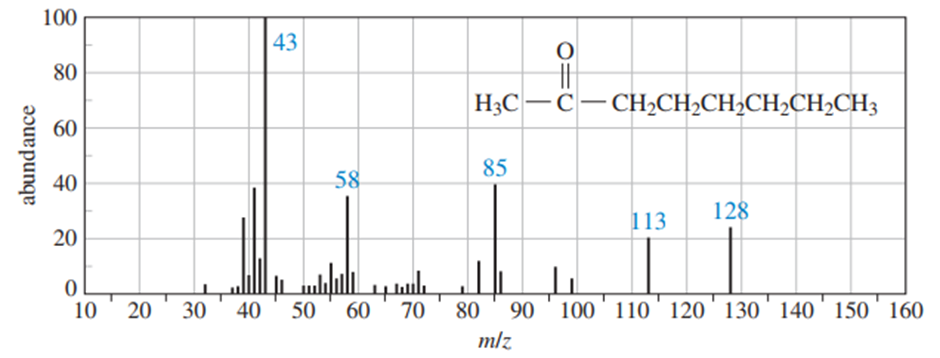
Use equations to show the fragmentation leading to each numbered peak in the mass spectrum of cotane-2-one.
Short Answer
 Fragmentation equations of octane-2-one
Fragmentation equations of octane-2-one
Learning Materials
Features
Discover
Chapter 18: Q4P (page 923)
Use equations to show the fragmentation leading to each numbered peak in the mass spectrum of cotane-2-one.

 Fragmentation equations of octane-2-one
Fragmentation equations of octane-2-one
All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the structures of the following derivatives.
(a) the 2,4-dinitrophenylhydrazone of acetone
(b) the semicarbazone of cyclopentanone
(c) cyclcobuanone oxime
(d) the ethylene acetal of hexan-2-one
(e) acetaldehyde diethyl acetal
(f) the ethyl hemiacetal of acetaldehyde
(g) the (Z) isomer of the ethyl imine of propiophenone
(h) the hemiacetal form of 6-hydroxyhexanal
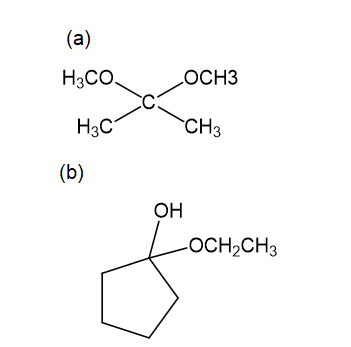
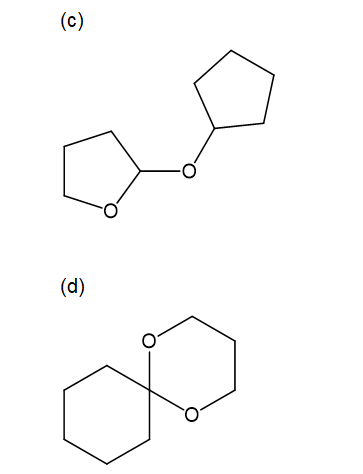
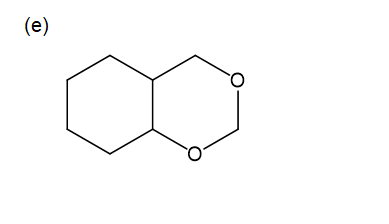
For each compound,
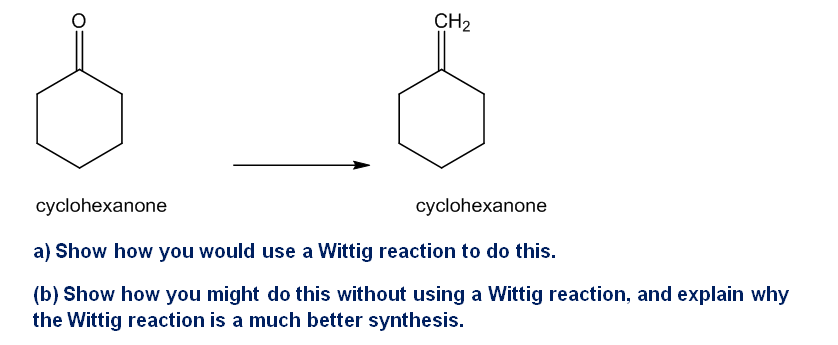
The Wittig reaction is useful for placing double bonds in less stable positions. For example, the following transformation is easily accomplished using a Wittig reaction
Show how you would synthesize each compound from starting materials containing no more than six carbon atoms.
(a)
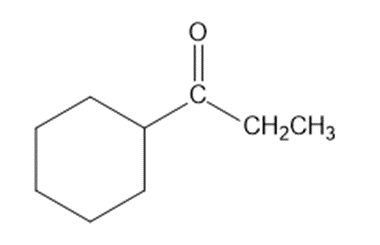
(b)
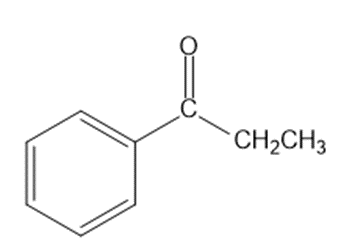
(c)
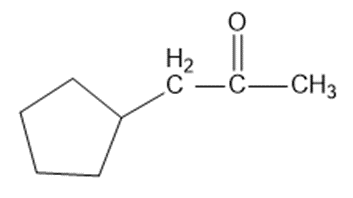
Show how the following transformations may be accomplished in good yield. You may use any additional reagents that are needed.
(a) bromobenzene → propiophenone
(b) CH3CH2CN → heptan-3-one
(c) benzoic acid → phenyl cyclopentyl ketone
(d) 1-bromo-hept-2-ene → oct-3- enal
What do you think about this solution?
We value your feedback to improve our textbook solutions.