Chapter 18: Q59SP (page 912)
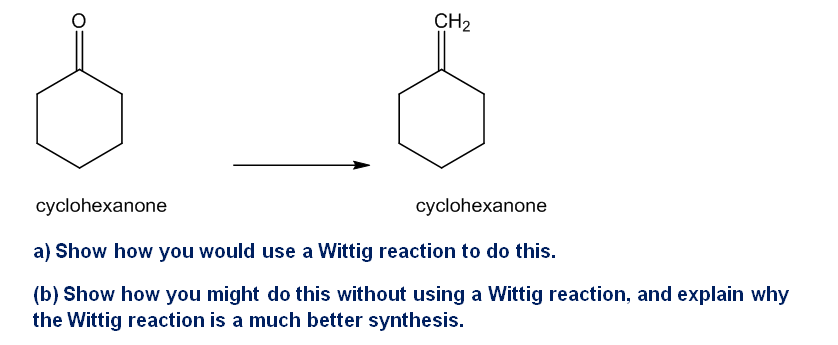
The Wittig reaction is useful for placing double bonds in less stable positions. For example, the following transformation is easily accomplished using a Wittig reaction
Short Answer
Learning Materials
Features
Discover
Chapter 18: Q59SP (page 912)
The Wittig reaction is useful for placing double bonds in less stable positions. For example, the following transformation is easily accomplished using a Wittig reaction


All the tools & learning materials you need for study success - in one app.
Get started for free
is frequently used for making derivatives of ketones and aldehydes because the products (, called derivatives) are even more likely than the phenlyhydrazones to be solids with sharp melting points. Propose a mechanism for the reaction of acetone with in a mildly acidic solution.
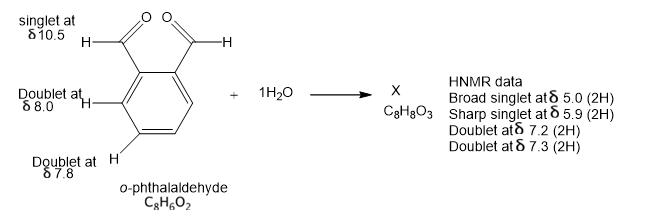
In the absence of water, ophthalaldehyde has the structure shown. Its strong IR absorption is at 1687 cm-1; the proton NMR data are shown by the structure. In the presence of water, a new compound is formed that has a strong IR absorption around 3400 cm-1 and no absortion in the C=0 region. propose a structure of X consistent with this information and suggest how X consistent with this information and suggest how X was formed.
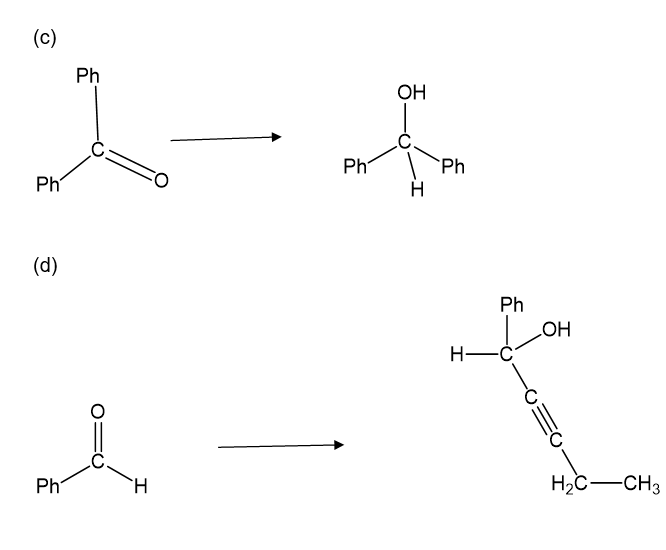
Show how you would accomplish the following syntheses.
(a).
(b).
(c).
(d).
Why were no products from McLafferty rearrangement observed in the spectrum of butan-2-one?
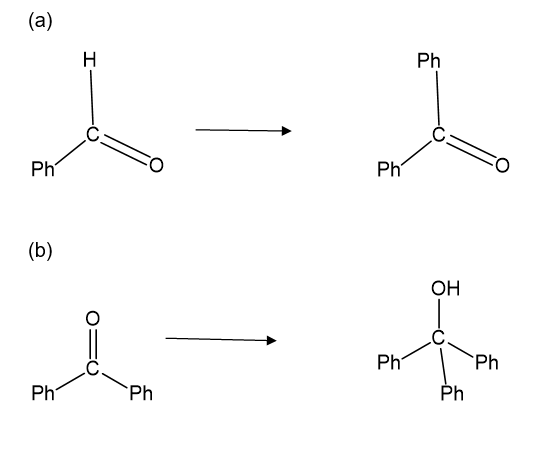
Show how you would accomplish the following synthetic conversions. You may use any additional reagents and solvents you need.
What do you think about this solution?
We value your feedback to improve our textbook solutions.