Chapter 18: Q11P (page 912)
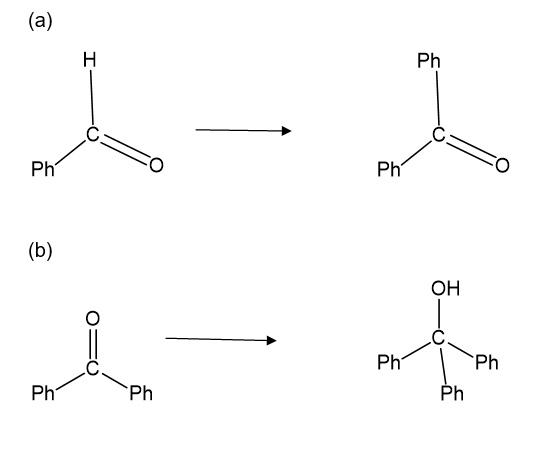
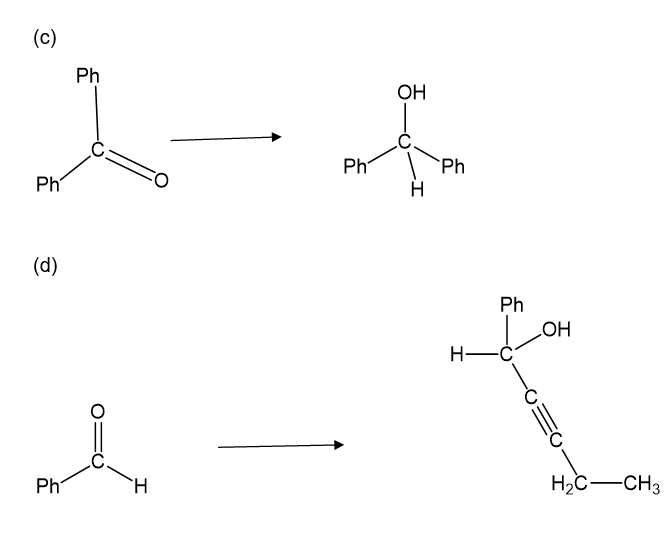
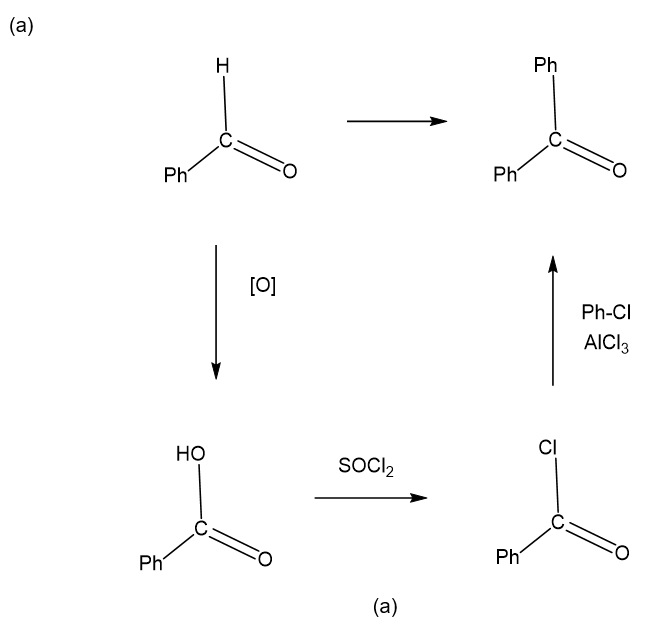
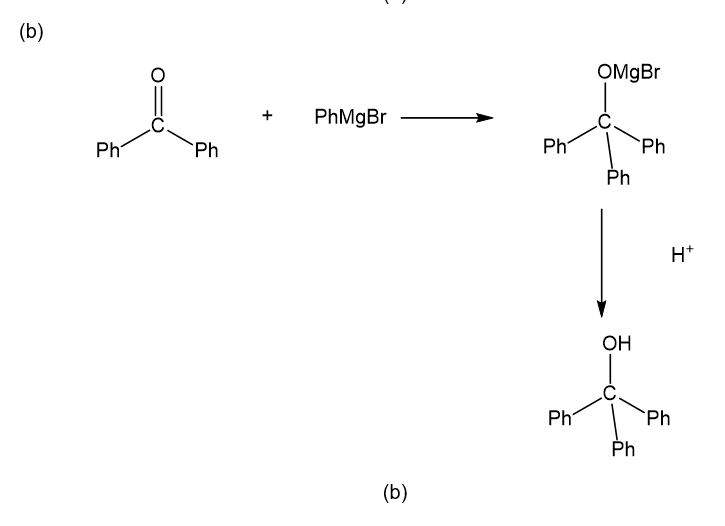
Show how you would accomplish the following synthetic conversions. You may use any additional reagents and solvents you need.
Short Answer
Learning Materials
Features
Discover
Chapter 18: Q11P (page 912)
Show how you would accomplish the following synthetic conversions. You may use any additional reagents and solvents you need.






All the tools & learning materials you need for study success - in one app.
Get started for free
Show how the following transformations may be accomplished in good yield. You may use any additional reagents that are needed.
(a) bromobenzene → propiophenone
(b) CH3CH2CN → heptan-3-one
(c) benzoic acid → phenyl cyclopentyl ketone
(d) 1-bromo-hept-2-ene → oct-3- enal
PROBLEM 18-10
Predict the products of the following reactions:
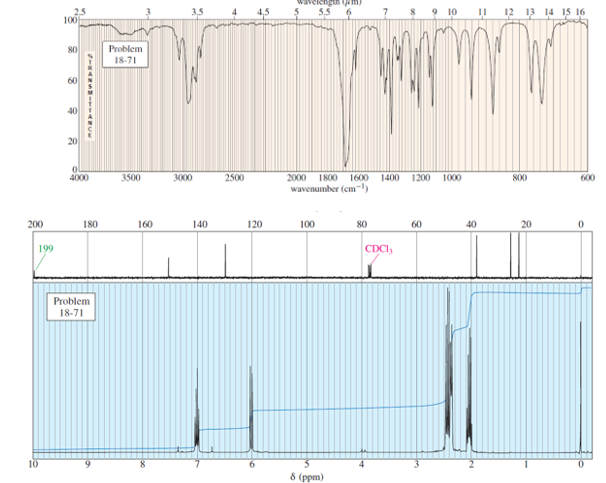
Question. The UV spectrum of an unknown compound shows values ofat 225 nm(E = 10,000) and at 318 nm(E = 40). The mass spectrum shows a molecular ion at m/z 96 and a prominent base peak at m/z68. The IR and NMR spectra follow. Propose a structure, and show how your structure corresponds to the observed absorptions. Propose a favorable fragmentation to account for the MS base peak at m/z 68 (loss of C2H4 ).
Use equations to show the fragmentation leading to each numbered peak in the mass spectrum of octane-2-one .
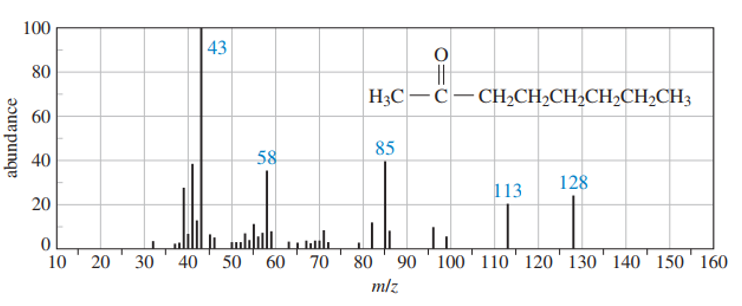
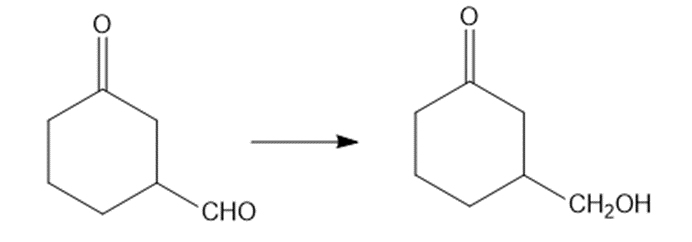
Show how would you accomplish the following syntheses. You may use whatever additional reagents you need.
(a)
(b)
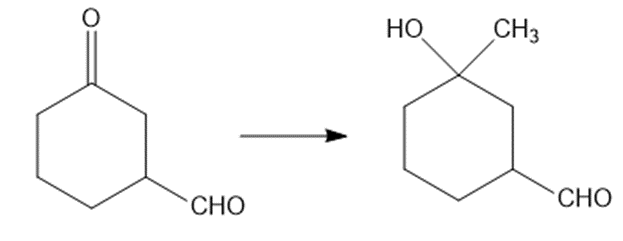
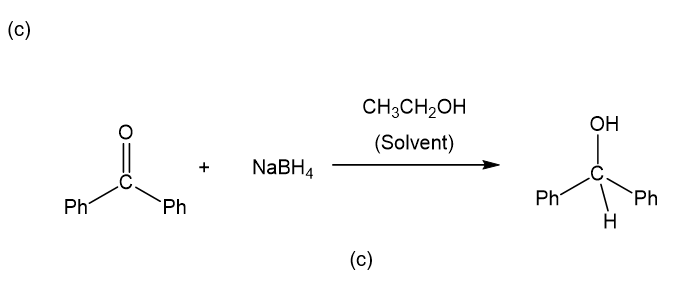
(c)
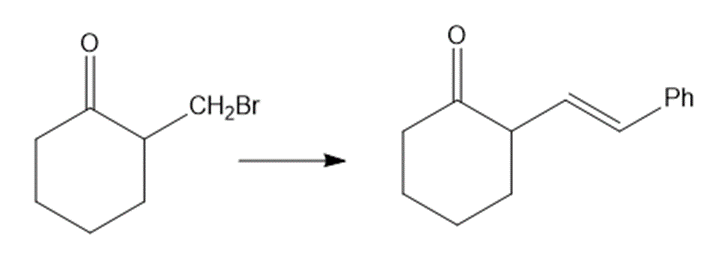
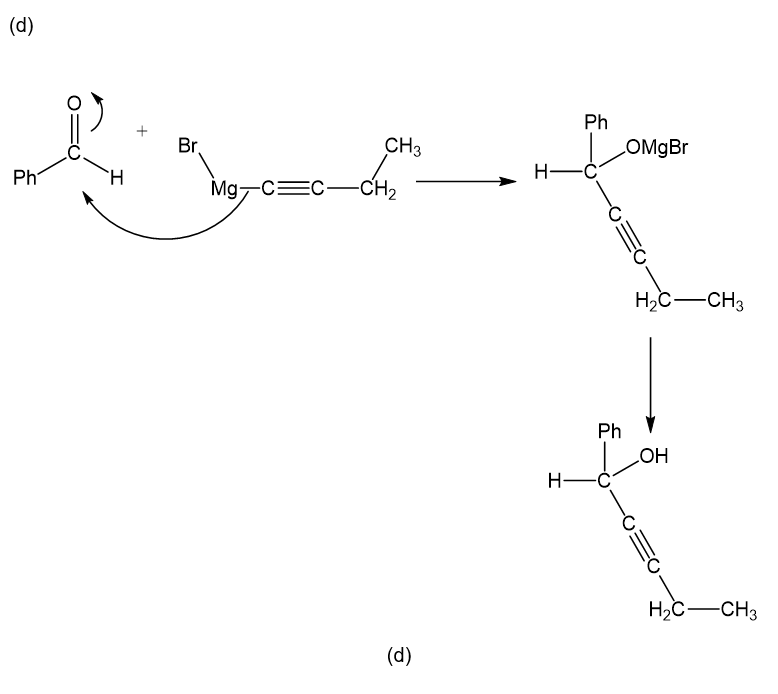
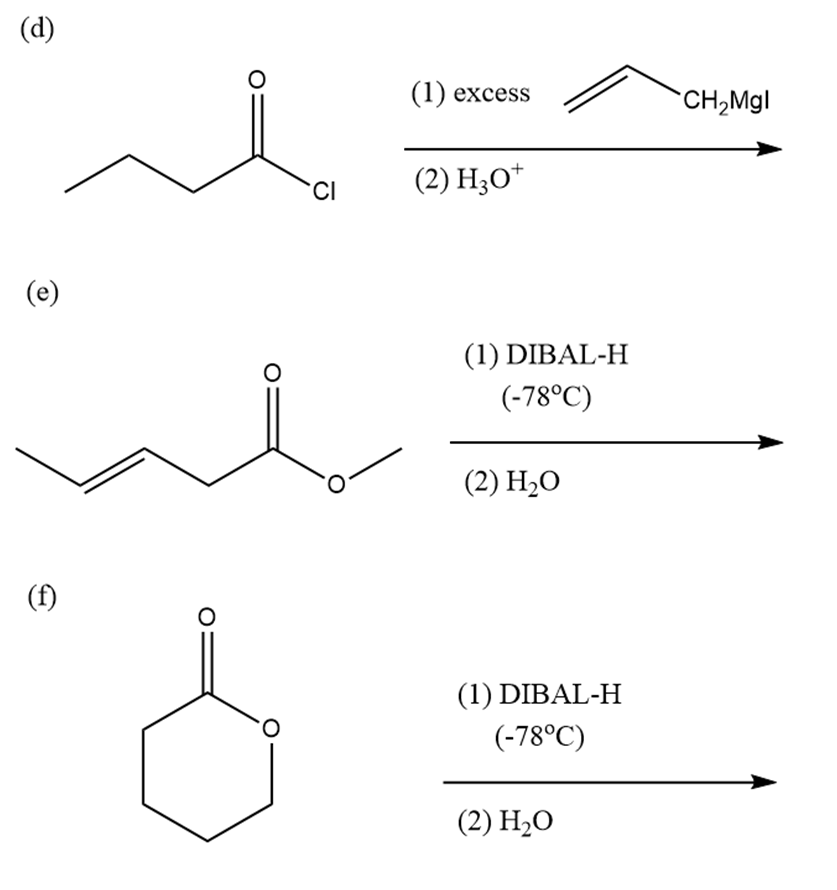
(d)
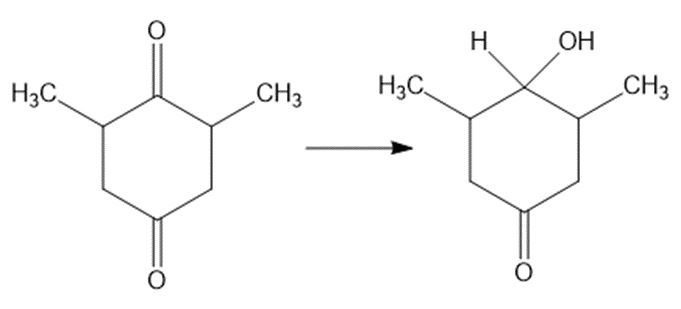
(e)
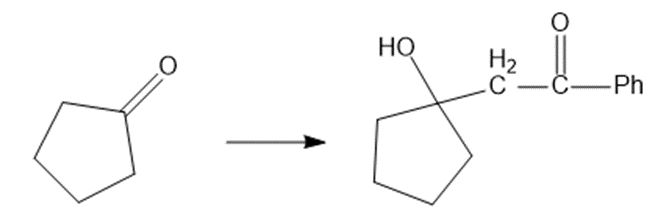
(f)
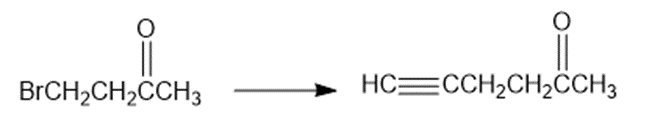
What do you think about this solution?
We value your feedback to improve our textbook solutions.