Chapter 18: Q29P (page 953)
Show how would you accomplish the following syntheses. You may use whatever additional reagents you need.
(a)
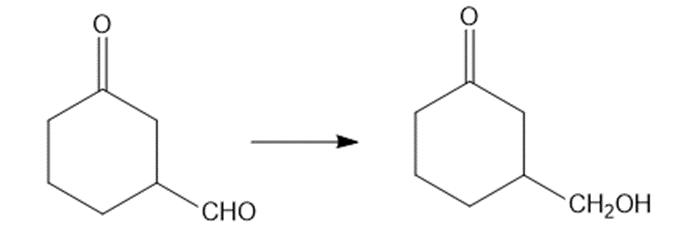
(b)
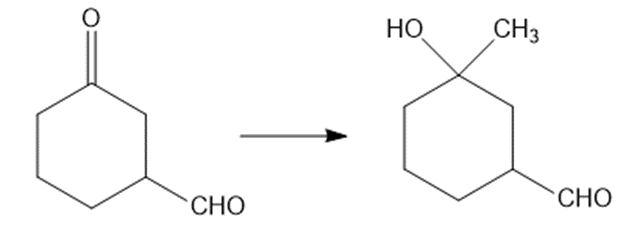
(c)
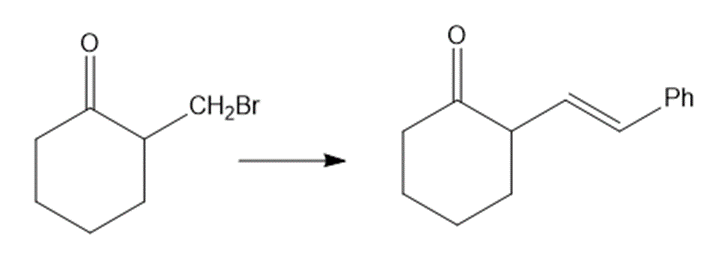
(d)
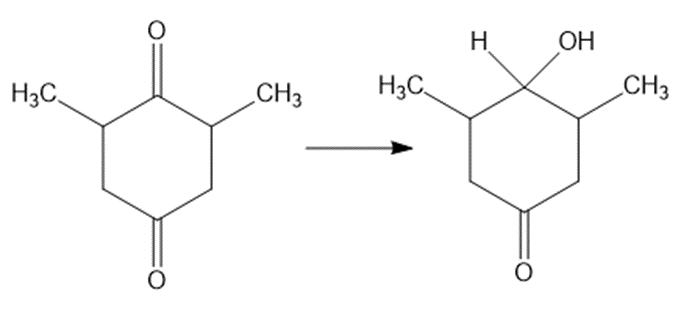
(e)

(f)
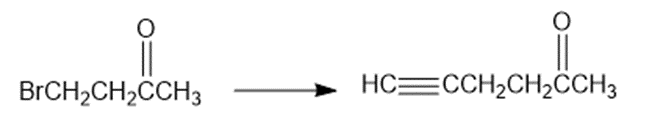
Short Answer
(a)
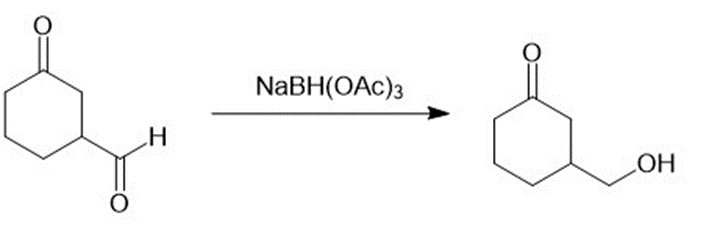
(b)

(c)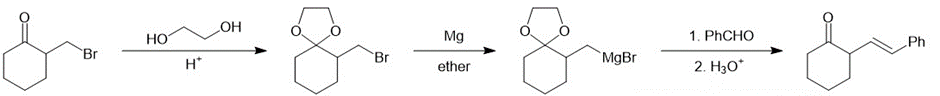
(d)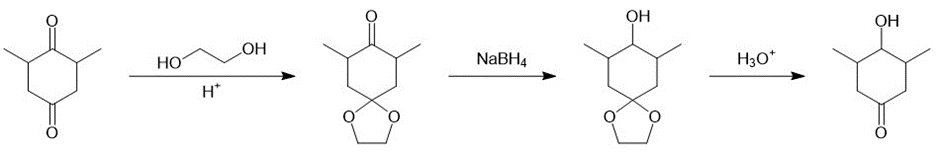
(e)
(f)

Learning Materials
Features
Discover
Chapter 18: Q29P (page 953)
Show how would you accomplish the following syntheses. You may use whatever additional reagents you need.
(a)

(b)

(c)

(d)

(e)

(f)

(a)

(b)

(c)
(d)
(e)
(f)

All the tools & learning materials you need for study success - in one app.
Get started for free
Show how you would accomplish the following syntheses.
(a).
(b).
(c).
(d).
Show how you would accomplish the following synthetic conversions by adding an organolithium reagent to an acid.
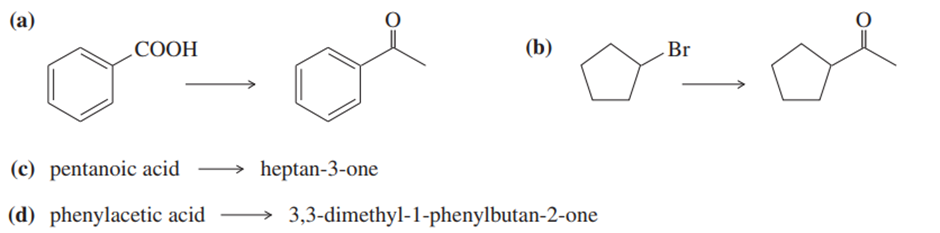
Show how the following transformations may be accomplished in good yield. You may use any additional reagents that are needed.
(a) bromobenzene → propiophenone
(b) CH3CH2CN → heptan-3-one
(c) benzoic acid → phenyl cyclopentyl ketone
(d) 1-bromo-hept-2-ene → oct-3- enal
(a) Propose a mechanism for the acid-catalyzed reaction of cyclohexanone with ethylene glycol to give cyclohexanone ethylene acetal.
(b) Propose a mechanism for the acid-catalyzed hydrolysis of cyclohexanone ethylene acetal.
(c) Compare the mechanisms you drew in parts (a) and (b). How similar are these mechanisms, comparing them in reverse order?
(d) Propose a mechanism for the acid-catalyzed hydrolysis of the acetal given in Problem 18-26(f).
Question: Propose mechanisms for the three imine-forming reactions just shown.
What do you think about this solution?
We value your feedback to improve our textbook solutions.