Chapter 18: Q17P. (page 944)
Question: Propose mechanisms for the three imine-forming reactions just shown.
Short Answer
Answer
(a)
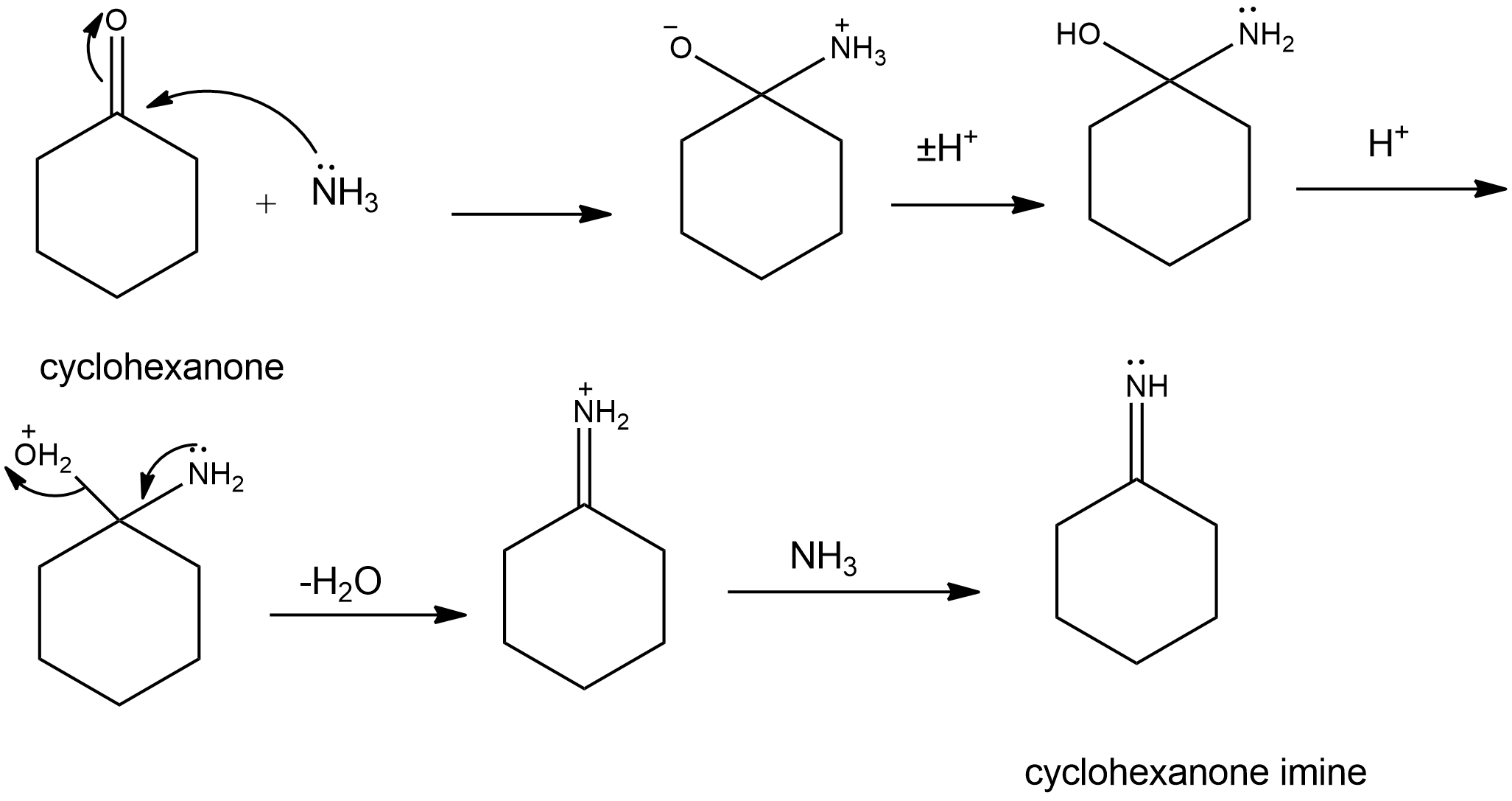
(b)
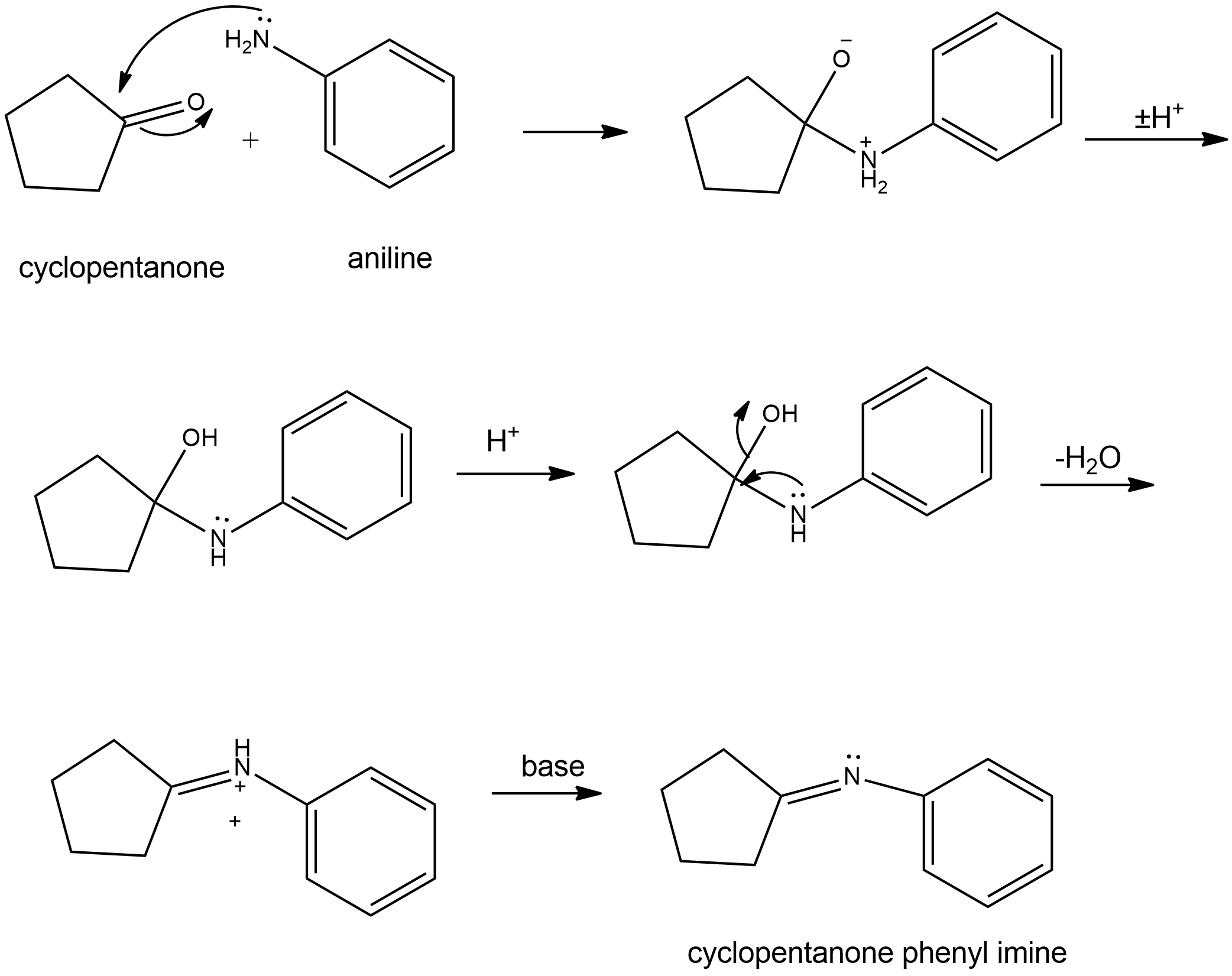
(c)
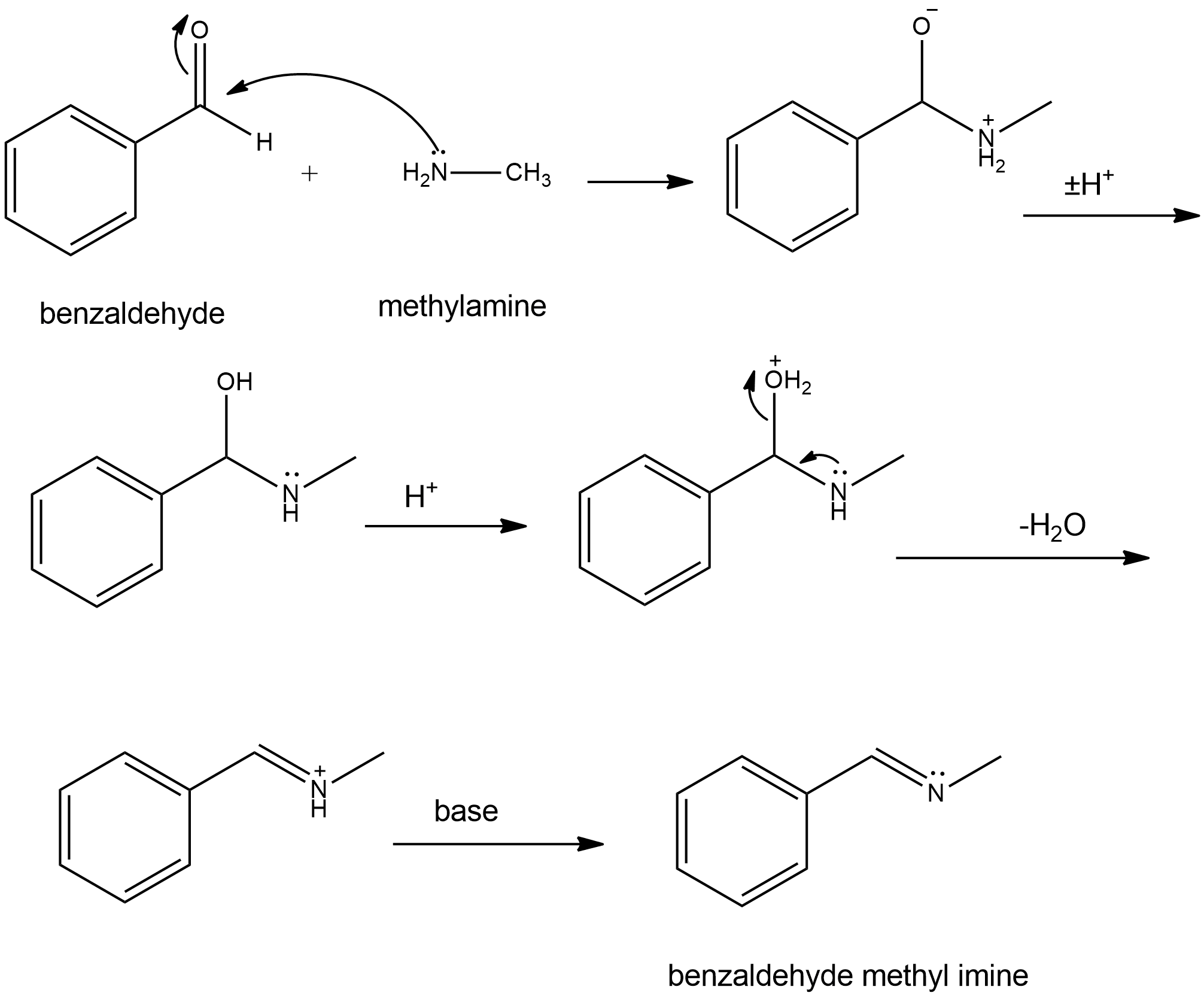
Learning Materials
Features
Discover
Chapter 18: Q17P. (page 944)
Question: Propose mechanisms for the three imine-forming reactions just shown.
Answer
(a)

(b)

(c)

All the tools & learning materials you need for study success - in one app.
Get started for free
Show how you would accomplish the following syntheses efficiently and in good yield. You may use any necessary reagents.
(a)
(b)
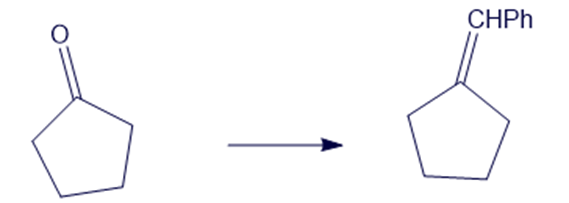
(c)
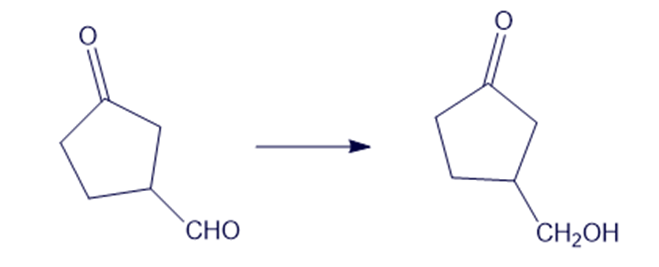
(d)
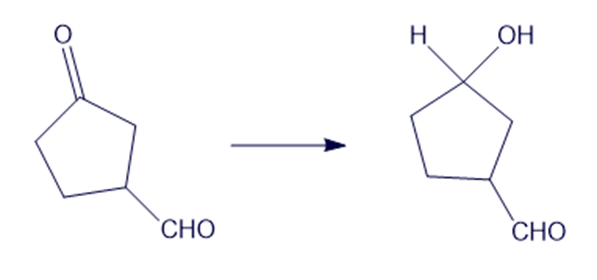
(e)
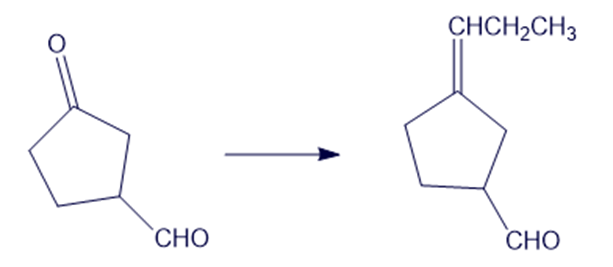
(f)
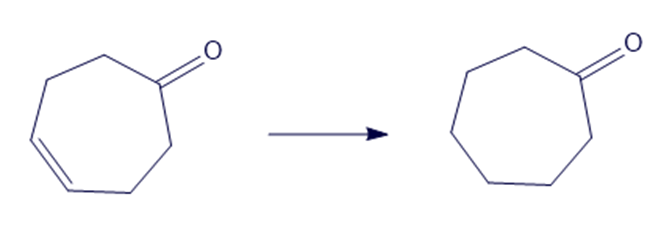
(g)

Propose a mechanism for both parts of the Wolff-Kishner reduction of cyclohexanone: the formation of the hydrazone, and then the base catalyzed reduction with evolution of nitrogen gas.
Show how you would synthesize the following derivatives from appropriate carbonyl compounds
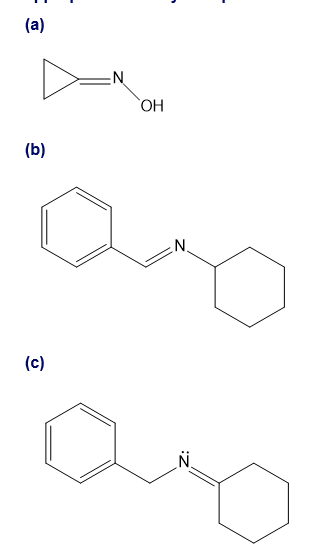
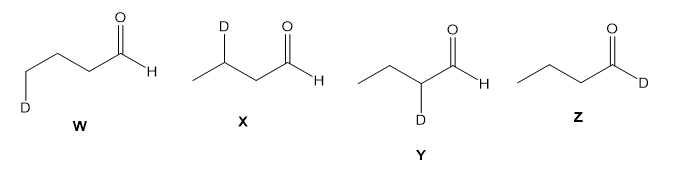
Assume you are a research physiologist trying to unravel a serious metabolic disorder. You have fed your lab animal Igor a deuterium-labelled substrate and now need to analyze the urinary metabolites. Show how you would differentiate these four deuterated aldehydes using mass spectrometry. Remember that deuterium has mass 2.
The family of macrolide antibiotics all have large rings (macrocycle) in which an ester is what makes the ring; a cyclic ester is termed as a lactone. One example is amphotericin B, used as an anti-fungal treatment of last resort because of its liver and heart toxicity. Professor Martin Burke of the University of Illnois has been making analogs to retain the antifungal properties but without the toxicity, including this structure published in 2015. (Nature Chemical Biology, (2015) doi: 10.1038/nchembio.1821). The carboxylate of amphotericin B has been replaced with the urea group (shown in red).
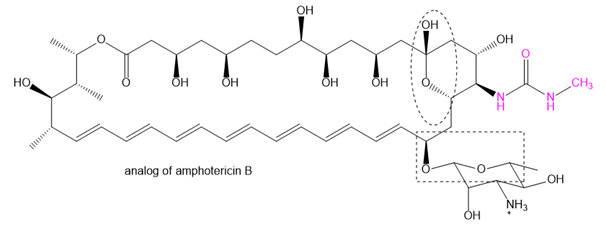
(a) Where is the lactone group that forms the ring?
(b) Two groups are circled. What type of functional group are they? Explain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.