Chapter 18: Q16P (page 942)
Show how you would accomplish the following synthesis.
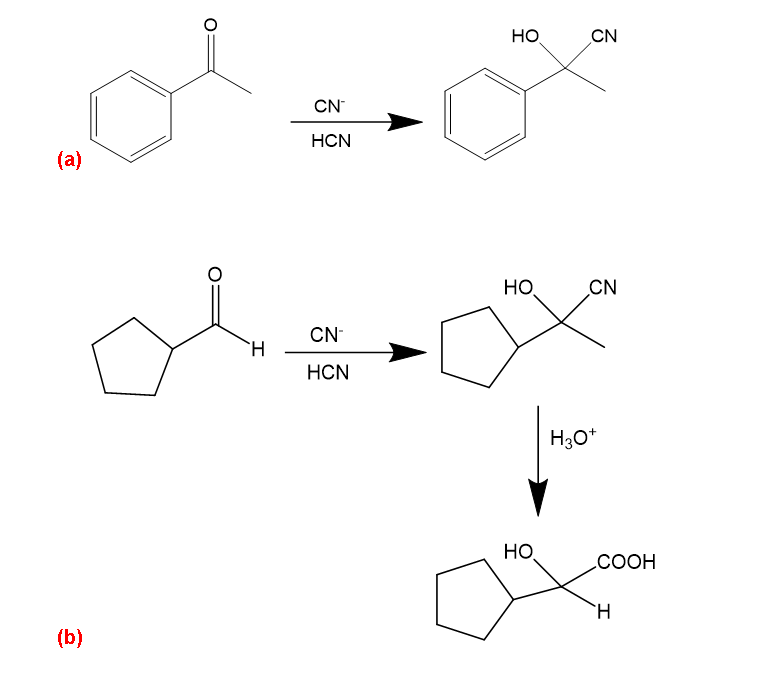
(a) Acetphenone→ acetophenone cyanohydrin
(b) Cyclopentancarbaldehyde→ 2-cyclopentyl-2-hydroxyacetic acid
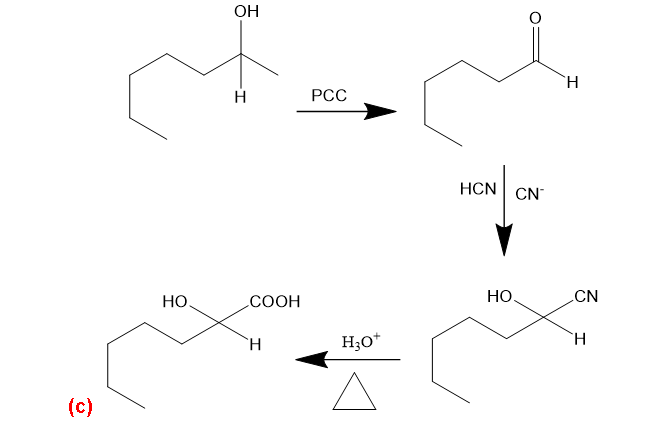
(c) Hexan-1-ol →2-hydroxyheptanoic acid
Short Answer
Learning Materials
Features
Discover
Chapter 18: Q16P (page 942)
Show how you would accomplish the following synthesis.
(a) Acetphenone→ acetophenone cyanohydrin
(b) Cyclopentancarbaldehyde→ 2-cyclopentyl-2-hydroxyacetic acid
(c) Hexan-1-ol →2-hydroxyheptanoic acid


All the tools & learning materials you need for study success - in one app.
Get started for free
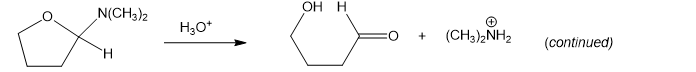
Question. (a) Simple aminoacetals hydrolyze quickly and easily in dilute acid. Propose a mechanism for hydrolysis of the following aminoacetal:
(b) The nucleosides that make up DNA have heterocyclic rings linked to deoxyribose by an aminoacetal functional group. Point out the aminoacetal linkages in deoxycytidine and deoxyadenosine.
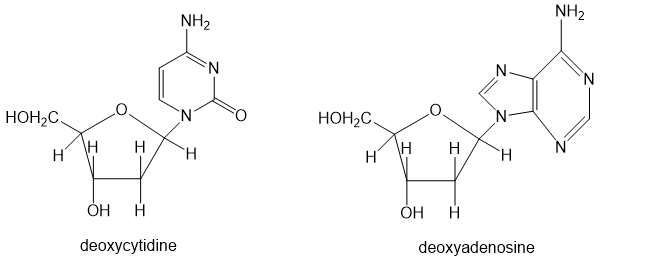
(c) The stability of our genetic code depends on the stability of DNA. We are fortunate that the aminoacetal linkages of DNA are not easily cleaved. Show why your mechanism for part (a) does not work so well with deoxycytidine and deoxyadenosine.
Rank the following carbonyl compounds in order of increasing equilibrium constant for hydration:
Question: The family of macrolide antibiotics all have large rings (macrocycle) in which an ester is what makes the ring; a cyclic ester is termed as a lactone. One example is amphotericin B, used as an anti-fungal treatment of last resort because of its liver and heart toxicity. Professor Martin Burke of the University of Illnois has been making analogs to retain the antifungal properties but without the toxicity, including this structure published in 2015. (Nature Chemical Biology, (2015) doi: 10.1038/nchembio.1821). The carboxylate of amphotericin B has been replaced with the urea group (shown in red).
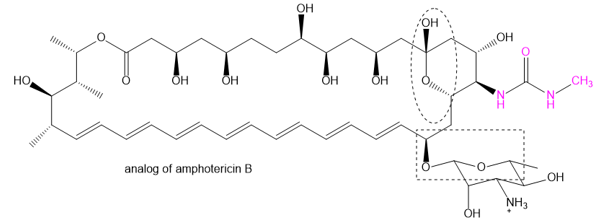
(a) Where is the lactone group that forms the ring?
(b) Two groups are circled. What type of functional group are they? Explain.
Show how you would accomplish the following syntheses efficiently and in good yield. You may use any necessary reagents.
(a)
(b)
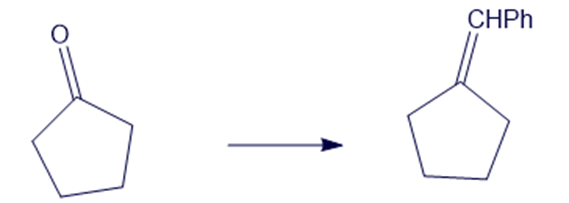
(c)
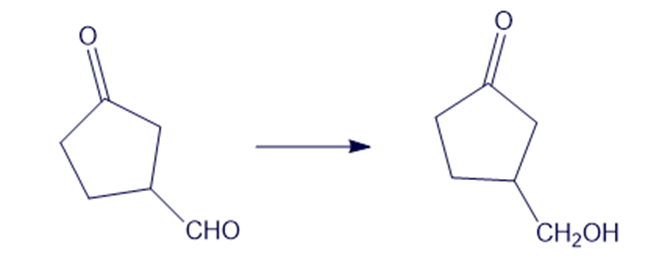
(d)
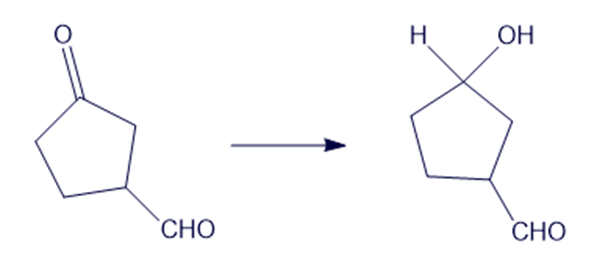
(e)
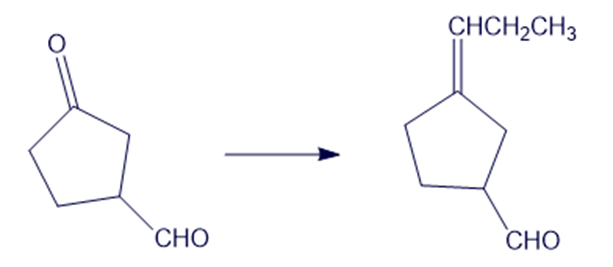
(f)
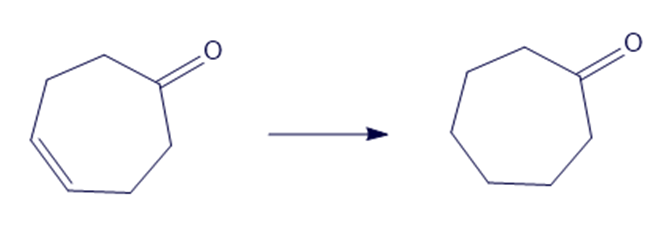
(g)

Use equations to show the fragmentation leading to each numbered peak in the mass spectrum of octane-2-one .
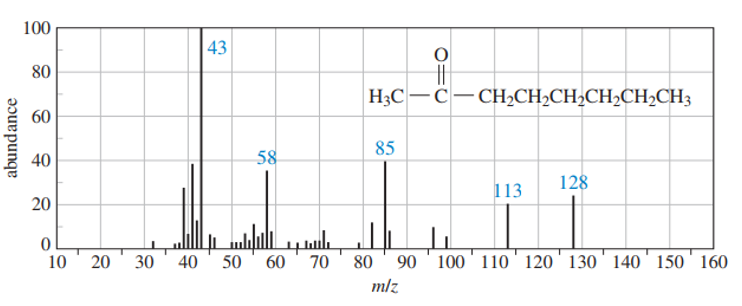
What do you think about this solution?
We value your feedback to improve our textbook solutions.