Chapter 18: Q40P (page 967)
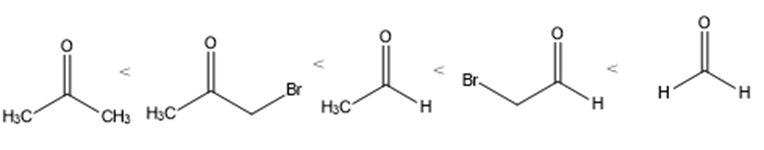
Rank the following carbonyl compounds in order of increasing equilibrium constant for hydration:
Short Answer
Learning Materials
Features
Discover
Chapter 18: Q40P (page 967)
Rank the following carbonyl compounds in order of increasing equilibrium constant for hydration:

All the tools & learning materials you need for study success - in one app.
Get started for free
Propose a mechanism for the acid-catalyzed reaction of benzaldehyde with methanol to give benzaldehyde dimethyl acetal.
Show how the following transformations may be accomplished in good yield. You may use any additional reagents that are needed.
(a) bromobenzene → propiophenone
(b) CH3CH2CN → heptan-3-one
(c) benzoic acid → phenyl cyclopentyl ketone
(d) 1-bromo-hept-2-ene → oct-3- enal
Hydration of alkynes (via oxymercuration) gives good yields of single compounds only with symmetrical or asymmetrical alkynes. Show what products would be from hydration fo each compound.
(a) hex-3-yne
(b) hex-2-yne
(c) hex-1-yne
(d) cyclodecyne
(e) 3-methylcyclodecyne
Sketch the expected proton NMR spectra of 3,3- dimethylbutanal.
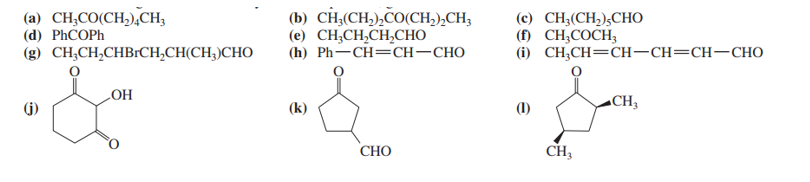
Name the following ketones and aldehydes. When possible, give both a common name and an IUPAC name.
What do you think about this solution?
We value your feedback to improve our textbook solutions.