Chapter 18: Q3P. (page 923)
Why were no products from McLafferty rearrangement observed in the spectrum of butan-2-one?
Short Answer
Butan-2-one has no gamma hydrogen, so no products were present in the McLafferty rearrangement.
Learning Materials
Features
Discover
Chapter 18: Q3P. (page 923)
Why were no products from McLafferty rearrangement observed in the spectrum of butan-2-one?
Butan-2-one has no gamma hydrogen, so no products were present in the McLafferty rearrangement.
All the tools & learning materials you need for study success - in one app.
Get started for free
Rank the following carbonyl compounds in order of increasing equilibrium constant for hydration:
PROBLEM 18-10
Predict the products of the following reactions:
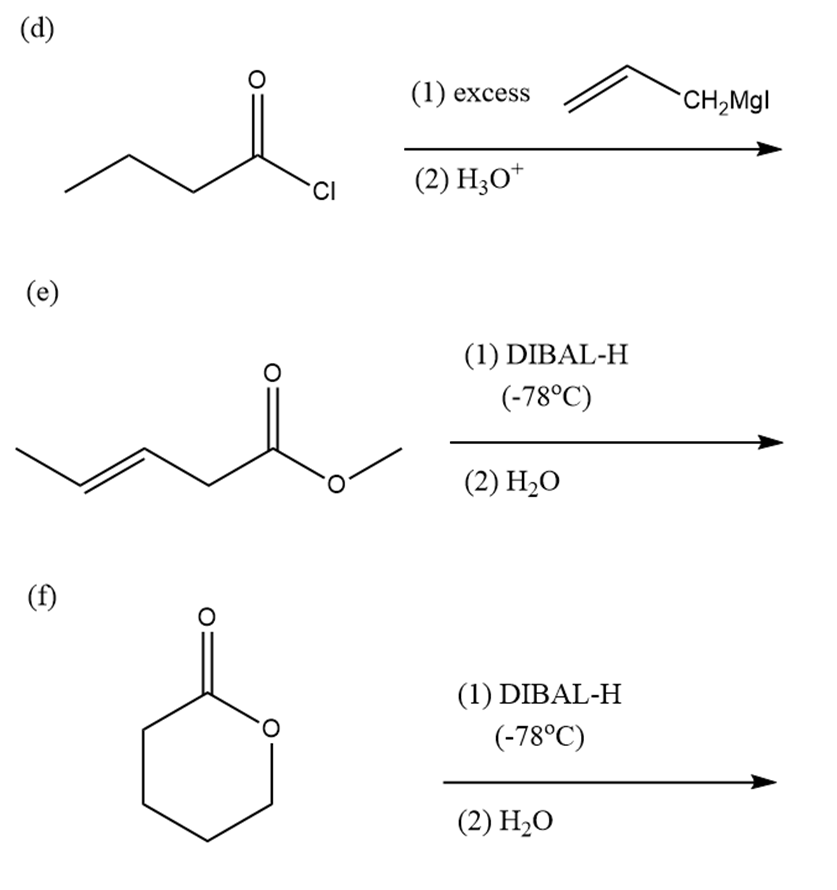
Name the following ketones and aldehydes. When possible, give both a common name and an IUPAC name.
(j) (k) (l)
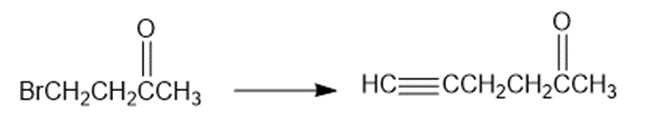
Show how would you accomplish the following syntheses. You may use whatever additional reagents you need.
(a)
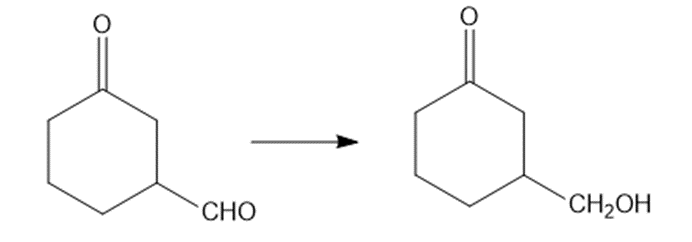
(b)
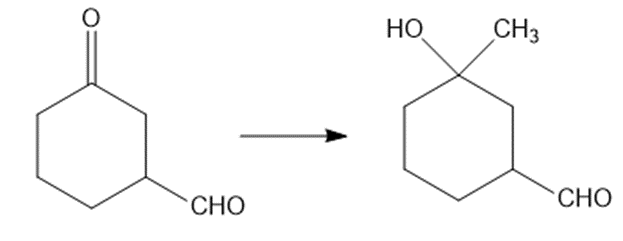
(c)
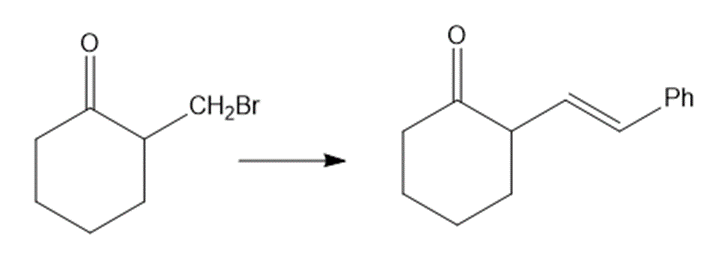
(d)
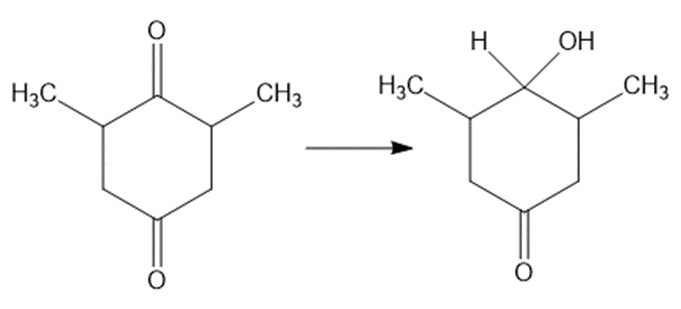
(e)
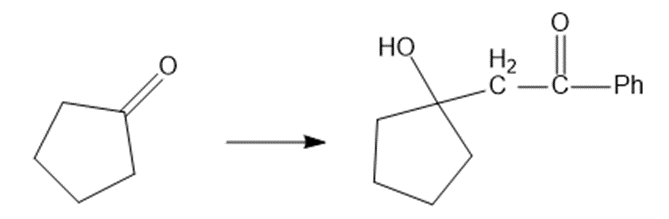
(f)
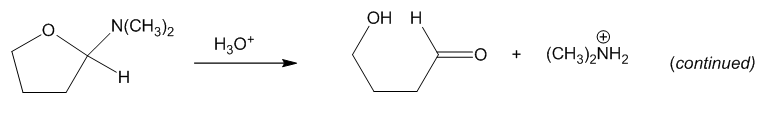
(a) Simple aminoacetals hydrolyze quickly and easily in dilute acid. Propose a mechanism for hydrolysis of the following aminoacetal:
(b) The nucleosides that make up DNA have heterocyclic rings linked to deoxyribose by an aminoacetal functional group. Point out the aminoacetal linkages in deoxycytidine and deoxyadenosine.
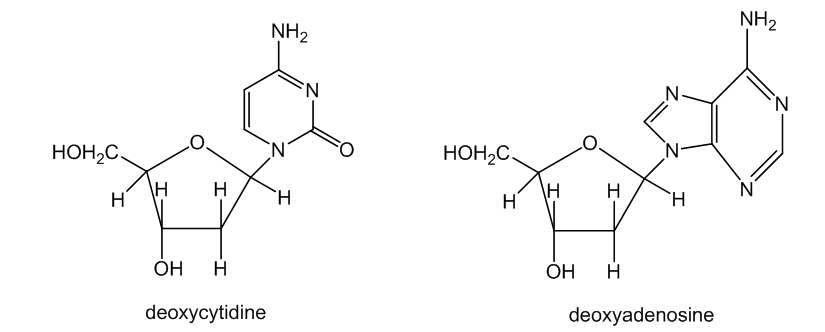
(c) The stability of our genetic code depends on the stability of DNA. We are fortunate that the aminoacetal linkages of DNA are not easily cleaved. Show why your mechanism for part (a) does not work so well with deoxycytidine and deoxyadenosine.
What do you think about this solution?
We value your feedback to improve our textbook solutions.