Chapter 18: Q38. (page 967)
Name the following ketones and aldehydes. When possible, give both a common name and an IUPAC name.
(j) (k) (l)
Short Answer
(a)
(b)
(c) heptanal
(d) benzophenone
(e)butanal
(f)
(g)
(h)
(i)
(j)
(k)
(l)
Learning Materials
Features
Discover
Chapter 18: Q38. (page 967)
Name the following ketones and aldehydes. When possible, give both a common name and an IUPAC name.
(j) (k) (l)
(a)
(b)
(c) heptanal
(d) benzophenone
(e)butanal
(f)
(g)
(h)
(i)
(j)
(k)
(l)
All the tools & learning materials you need for study success - in one app.
Get started for free
Question. The mass spectrum of unknown compound A shows a molecular ion at m/z 116 and prominent peaks at m/z 87 and m/z 101. Its UV spectrum shows no maximum above 200 nm. The IR and NMR spectra of A follow. When A is washed with dilute aqueous acid, extracted into dichloromethane , and the solvent evaporated, it gives a product B. B shows a strong carbonyl signal at 1715 cm-1in the IR spectrum and a weak maximum at 274nm(E =16) in the UV spectrum. The mass spectrum of B shows a molecular ion of m/z 72. Determine the structures of A and B, and show the fragmentation to account for the peaks at m/z 87 and 101.


Propose a mechanism for the acid-catalyzed reaction of benzaldehyde with methanol to give benzaldehyde dimethyl acetal.
Show what alcohols and carbonyl compounds give the following derivatives.
(a)
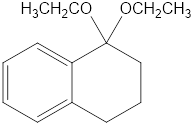
(b)
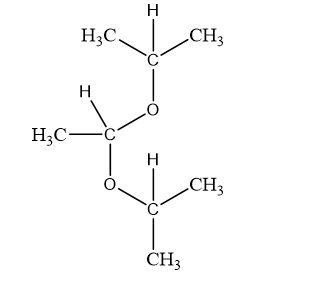
(c)
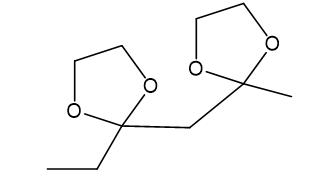
(d)
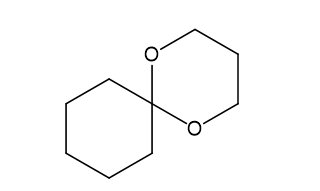
(e)
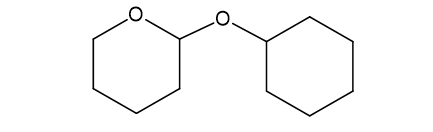
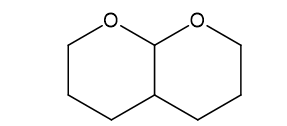
(f)
Show how the following transformations may be accomplished in good yield. You may use any additional reagents that are needed.
(a) bromobenzene → propiophenone
(b) CH3CH2CN → heptan-3-one
(c) benzoic acid → phenyl cyclopentyl ketone
(d) 1-bromo-hept-2-ene → oct-3- enal
Sodium triacetoxyborohydride, , is a mild reducing agent that reduces aldehydes more quickly than ketones. It can be used to reduce aldehydes in the presence of ketones, such as in the following reaction:

(a) Draw a complete Lewis structure for sodium triacetoxyborohydride.
(b) Propose a mechanism for the reduction of an aldehyde by sodium triacetoxyborohydride.
What do you think about this solution?
We value your feedback to improve our textbook solutions.