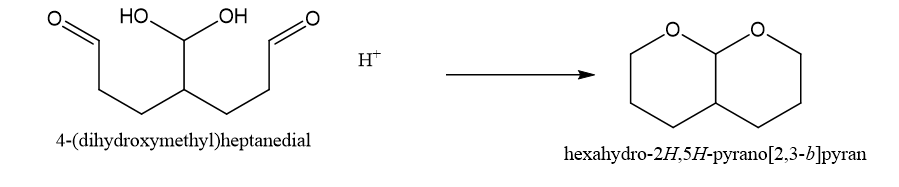
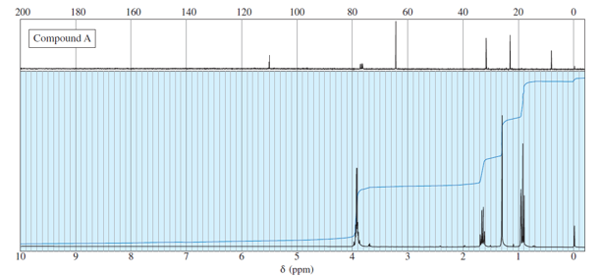
Chapter 18: Q26P (page 950)
Show what alcohols and carbonyl compounds give the following derivatives.
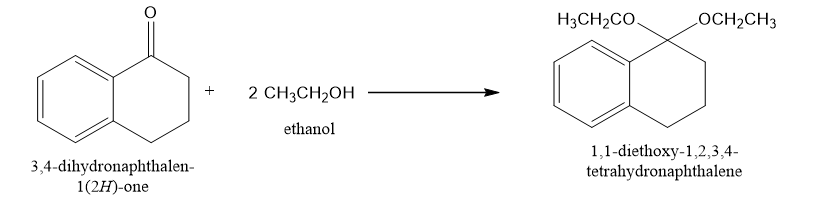
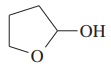
(a)
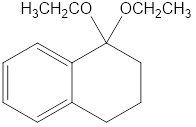
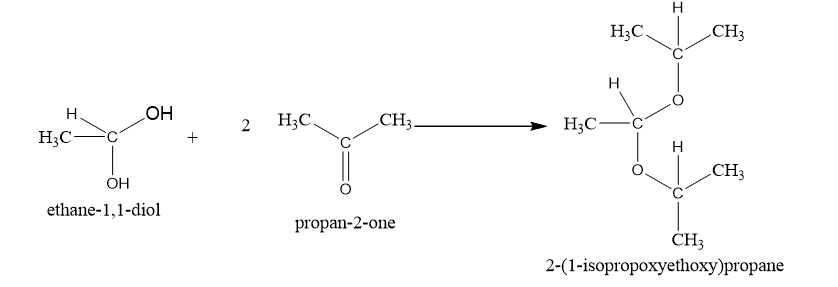
(b)

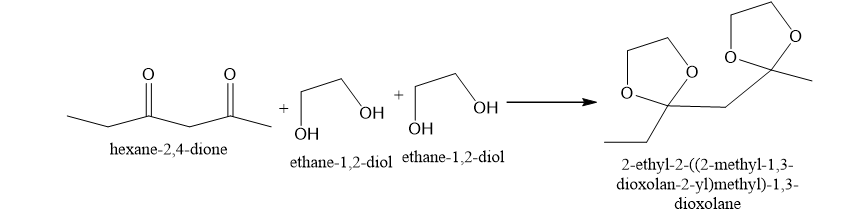
(c)
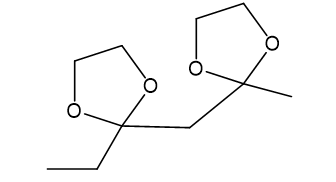
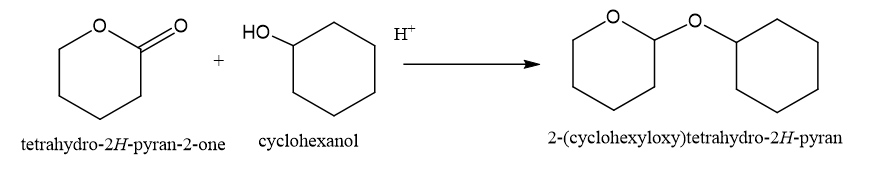
(d)
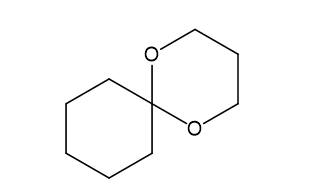
(e)

(f)
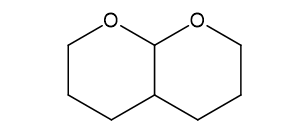
Short Answer
(a)Generally in presence of base like TsOH this is formed.
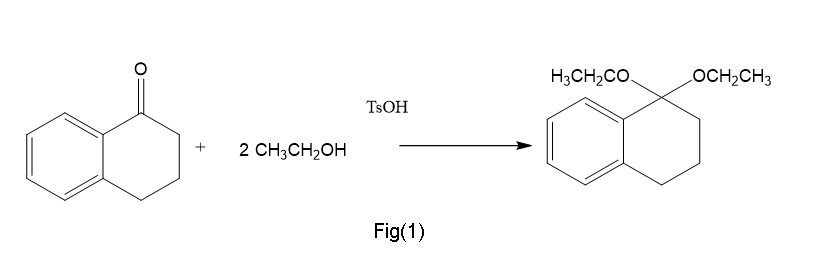
(b)This is obtained in presence of H+,
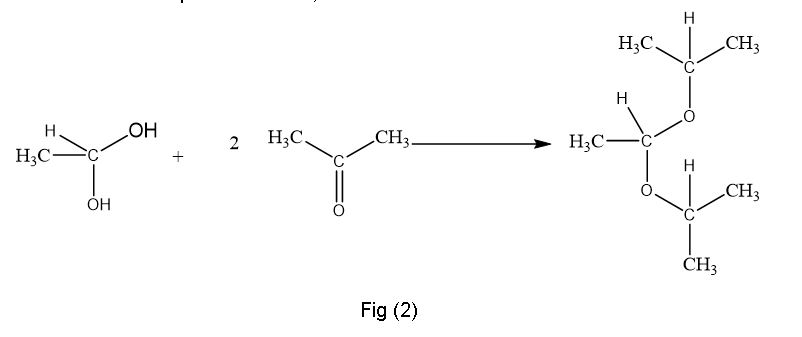
(c)This is produced in presence acid catalyst like H+,
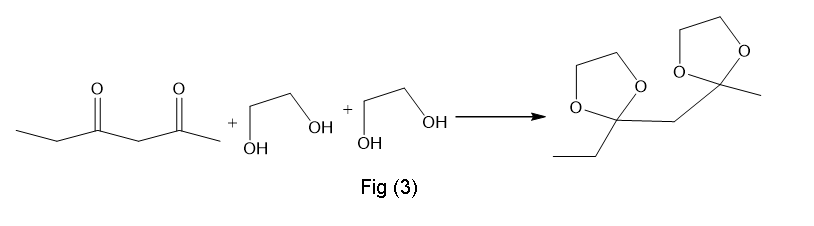
(d)This is also formed in presence of H+,
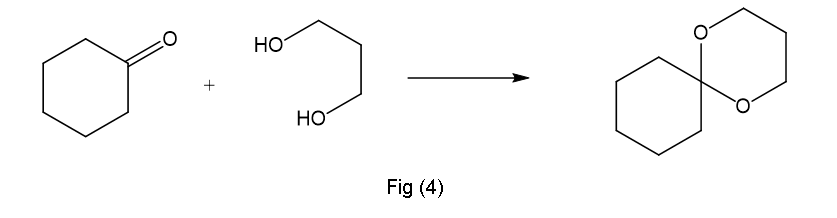
(e)This is formed in presence of H+,
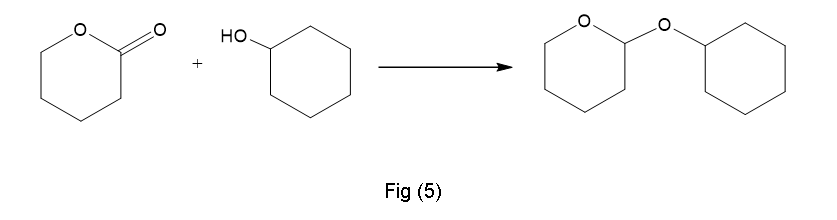
(f) This is results in presence of H+,
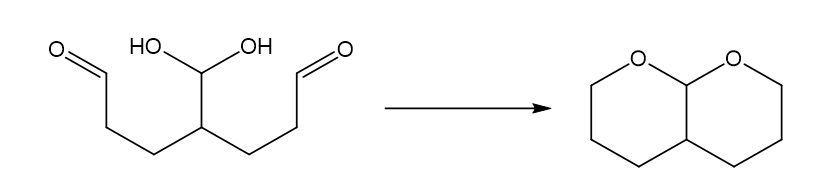
Fig (6)