Chapter 18: Q25. (page 950)
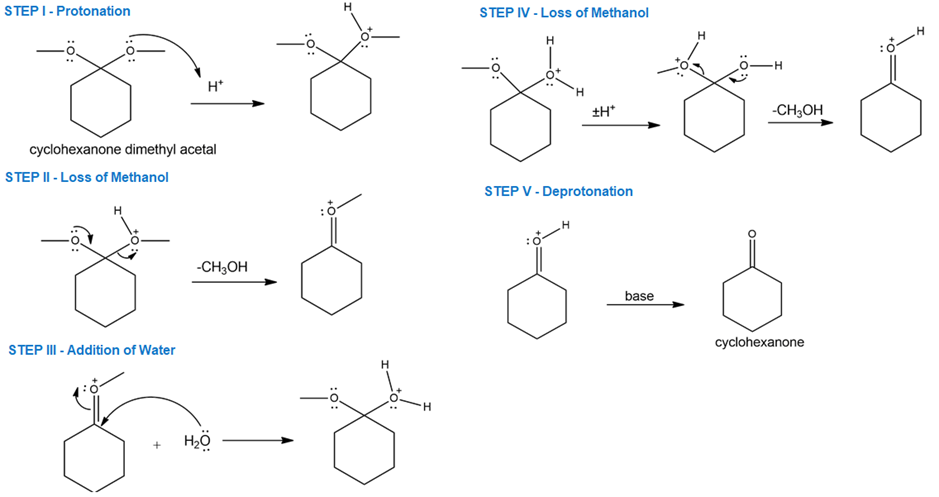
Propose a mechanism for the acid-catalyzed hydrolysis of cyclohexanone dimethyl acetal.
Short Answer
Dilute aqueous acid can be used for the hydrolysis of cyclohexanone dimethyl acetal. The mechanism is shown below.
Learning Materials
Features
Discover
Chapter 18: Q25. (page 950)
Propose a mechanism for the acid-catalyzed hydrolysis of cyclohexanone dimethyl acetal.
Dilute aqueous acid can be used for the hydrolysis of cyclohexanone dimethyl acetal. The mechanism is shown below.

All the tools & learning materials you need for study success - in one app.
Get started for free
Propose a mechanism for each cyanohydrin synthesis just shown.
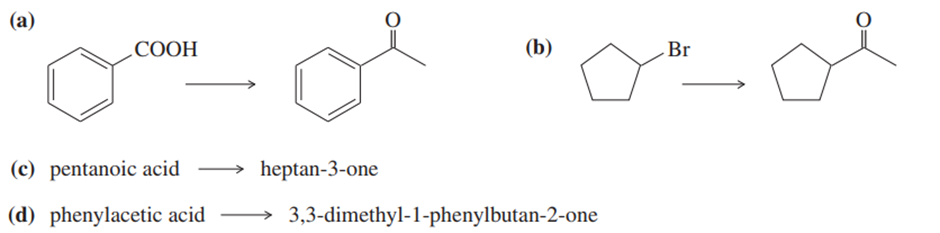
Show how you would accomplish the following syntheses.
(a).
(b).
(c).
(d).
Show how the following transformations may be accomplished in good yield. You may use any additional reagents that are needed.
(a) bromobenzene → propiophenone
(b) CH3CH2CN → heptan-3-one
(c) benzoic acid → phenyl cyclopentyl ketone
(d) 1-bromo-hept-2-ene → oct-3- enal
Show how you would accomplish the following synthetic conversions by adding an organolithium reagent to an acid.
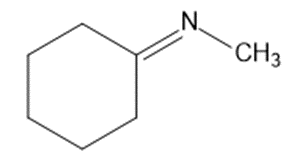
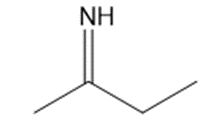
Give the structures of the carbonyl compound and the amine used to form the following imines.
(a)
(b)
(c)
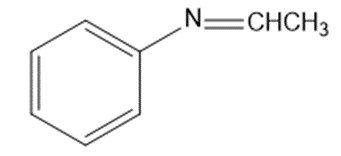
(d)
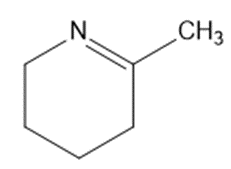
(e)
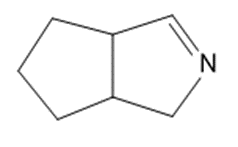
(f)
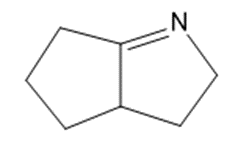
What do you think about this solution?
We value your feedback to improve our textbook solutions.