Chapter 18: Q42P (page 967)
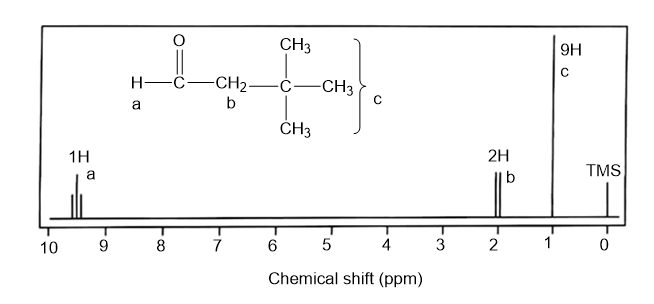
Sketch the expected proton NMR spectrum of 3,3-dimethylbutanal.
Short Answer
The expected proton NMR spectrum of 3,3-dimethylbutanal is as follows:
Learning Materials
Features
Discover
Chapter 18: Q42P (page 967)
Sketch the expected proton NMR spectrum of 3,3-dimethylbutanal.
The expected proton NMR spectrum of 3,3-dimethylbutanal is as follows:

All the tools & learning materials you need for study success - in one app.
Get started for free
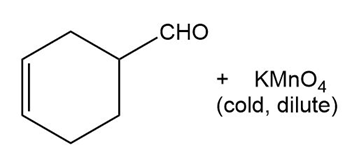
Predict the major products of the following reactions.
(a) (b)

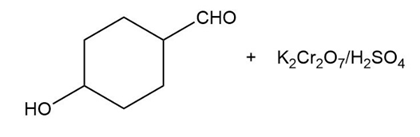
(c) (d)

Predict the products formed when cyclopentanone reacts with the following reagents.
(a) CH3NH2 , H+
(b) excess CH3OH , H+
(c) hydroxylamine and weak acid
(d) ethylene glycol and p-toluenesulfuric acid
(e) phenylhydrazine and weak acid
(f) PhMgBr and then mild H3O+
(g) Tollens reagent
(h) sodium acetlylide, then H3O+
(i) hydrazine, then hot. fused KOH
(j) Ph3P=CH2
(k) sodium cyanide
(l) acidic hydrolysis of the product from (k)
Draw the structures of the following derivatives.
(a) the 2,4-dinitrophenylhydrazone of acetone
(b) the semicarbazone of cyclopentanone
(c) cyclcobuanone oxime
(d) the ethylene acetal of hexan-2-one
(e) acetaldehyde diethyl acetal
(f) the ethyl hemiacetal of acetaldehyde
(g) the (Z) isomer of the ethyl imine of propiophenone
(h) the hemiacetal form of 6-hydroxyhexanal
Question. An unknown compound gives a molecular ion of m/z 70 in the mass spectrum. It reacts with semicarbazide hydrochloride to give a crystalline derivative, but it gives a negative Tollens test. The NMR and IR spectra follow. Propose a structure for this compound, and give peak assignments to account for the absorptions in spectra. Explain why the signal atin the IR spectrum appears at an unusual frequency.
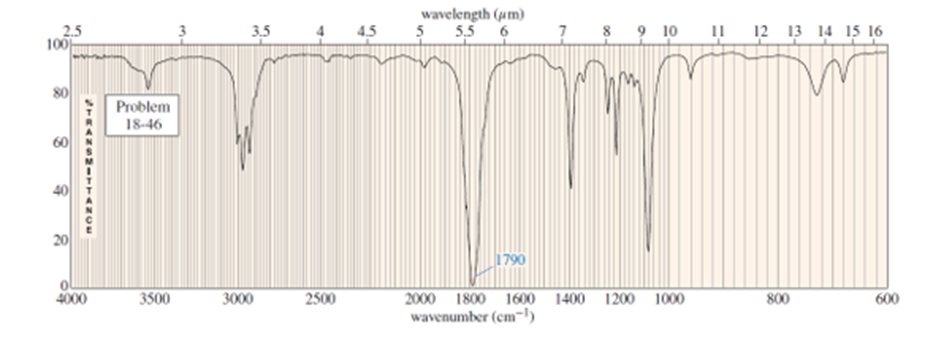
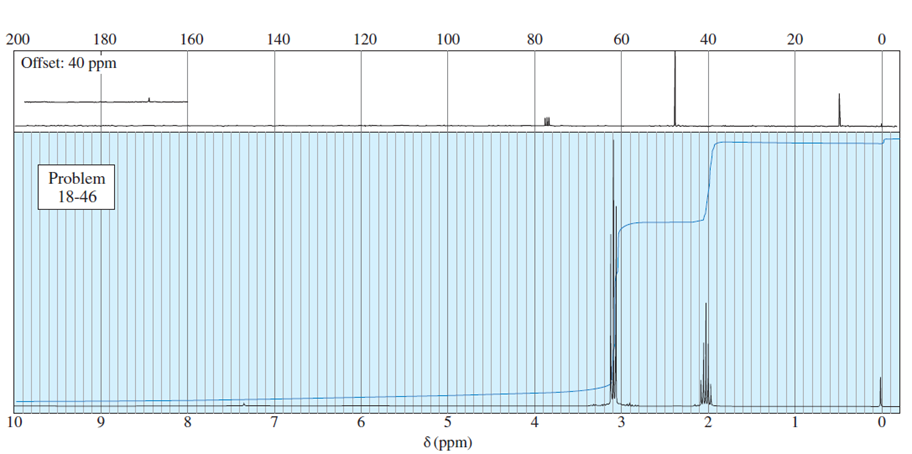
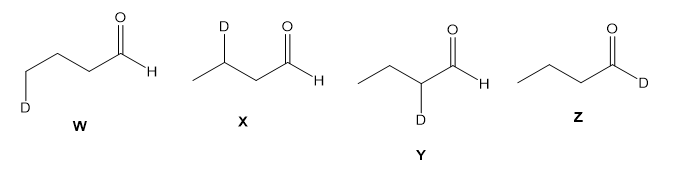
Assume you are a research physiologist trying to unravel a serious metabolic disorder. You have fed your lab animal Igor a deuterium-labelled substrate and now need to analyze the urinary metabolites. Show how you would differentiate these four deuterated aldehydes using mass spectrometry. Remember that deuterium has mass 2.
What do you think about this solution?
We value your feedback to improve our textbook solutions.