Chapter 18: Q14. (page 940)
Rank the following compounds in order of increasing amount of hydrate present at equilibrium.
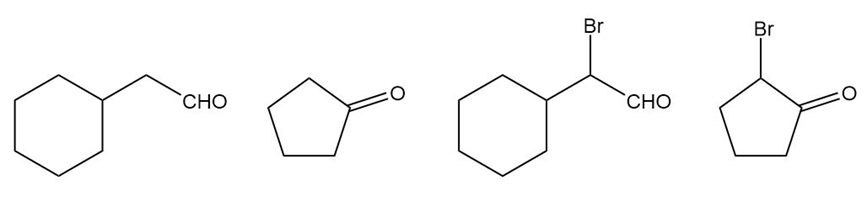
Short Answer
The compounds in the increasing order amount of hydrate present at equilibrium is shown below.
Learning Materials
Features
Discover
Chapter 18: Q14. (page 940)
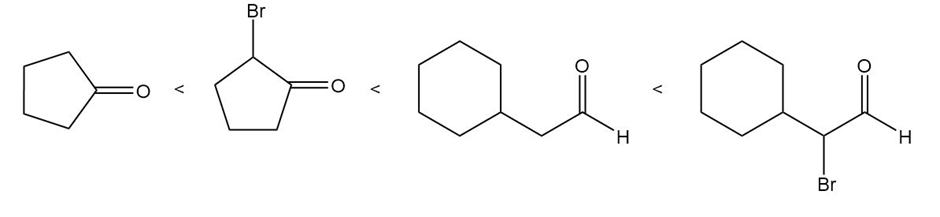
Rank the following compounds in order of increasing amount of hydrate present at equilibrium.

The compounds in the increasing order amount of hydrate present at equilibrium is shown below.

All the tools & learning materials you need for study success - in one app.
Get started for free
Two structures for the sugar glucose are shown on page 950. Interconversion of the open-chain and cyclic hemiacetal forms is catalyzed by either acid or base.
(a) Propose a mechanism for the cyclization, assuming a trace of acid is present.
(b) The cyclic hemiacetal is more stable than the open-chain form, so very little of the open-chain form is present at equilibrium. Will an aqueous solution of glucose reduce Tollens reagent and give a positive Tollens test? Explain.
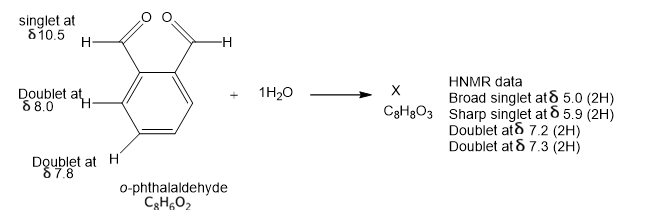
In the absence of water, ophthalaldehyde has the structure shown. Its strong IR absorption is at 1687 cm-1; the proton NMR data are shown by the structure. In the presence of water, a new compound is formed that has a strong IR absorption around 3400 cm-1 and no absortion in the C=0 region. propose a structure of X consistent with this information and suggest how X consistent with this information and suggest how X was formed.
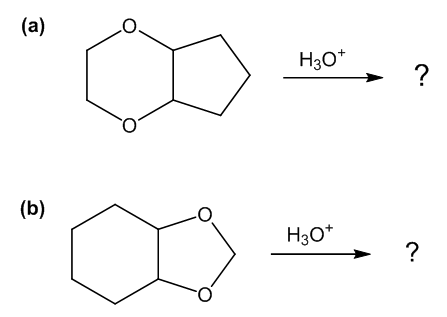
One of these reacts with dilute aqueous acid and the other does not. Give a mechanism for the one that reacts and show why this mechanism does not work for the other one.
Propose mechanisms for
(a) the acid-catalyzed hydration of chloral to form chloral hydrate.
(b) the base-catalyzed hydration of acetone to form acetone hydrate
Question: Propose mechanisms for the three imine-forming reactions just shown.
What do you think about this solution?
We value your feedback to improve our textbook solutions.