Chapter 18: Q22P (page 947)
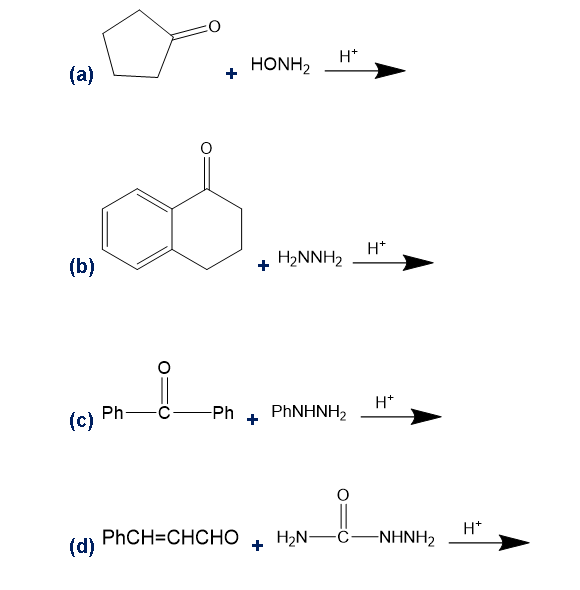
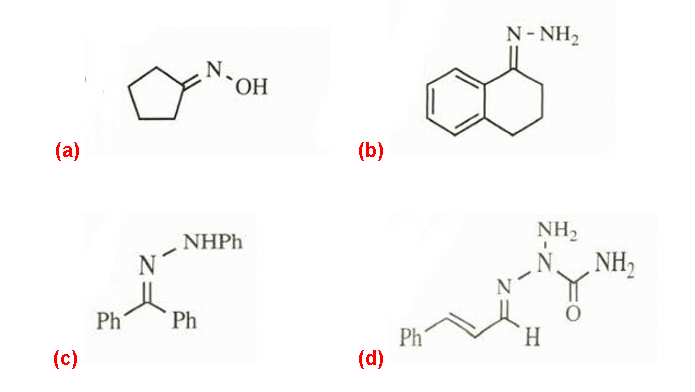
Predict the products of the following reactions.
Short Answer
Learning Materials
Features
Discover
Chapter 18: Q22P (page 947)
Predict the products of the following reactions.


All the tools & learning materials you need for study success - in one app.
Get started for free
Trimethylphosphine is a stronger nucleophile than triphenylphosphine, but it is rarely used to make ylides. Why is trimethylphosphine unsuitable for making most phosphorus ylides?
In the mechanism for acetal hydrolysis shown, the ring oxygen atom was protonated first, the ring was cleaved, and then the methoxy group was lost. The mechanism could also be written to show the methoxy oxygen protonating and cleaving first, followed by ring cleavage. Draw the alternative mechanism.
Show how you would synthesize each compound from starting materials containing no more than six carbon atoms.
(a)
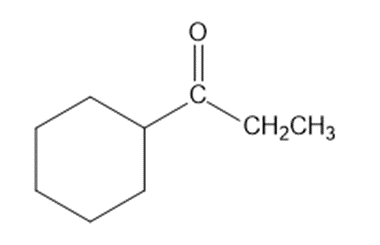
(b)
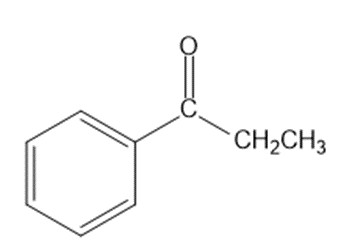
(c)
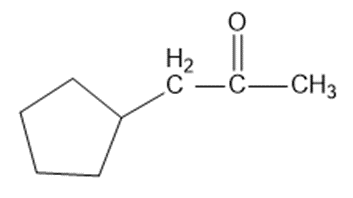
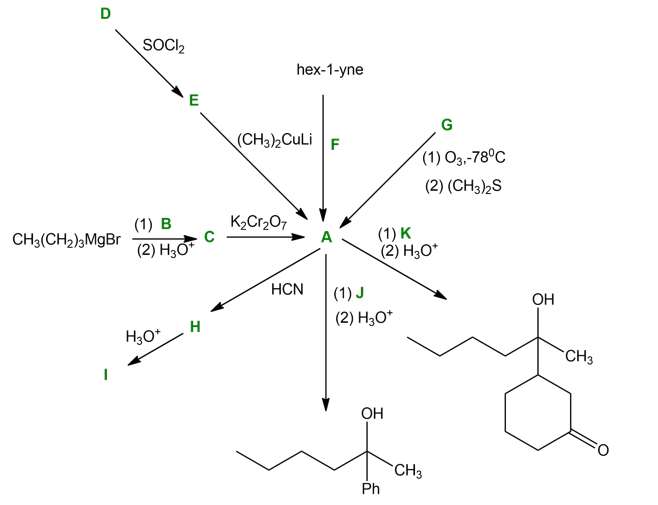
Question: Solving the following road-map problem depends on determining the structure of A, the key intermediate. Give structures for compounds A through K.
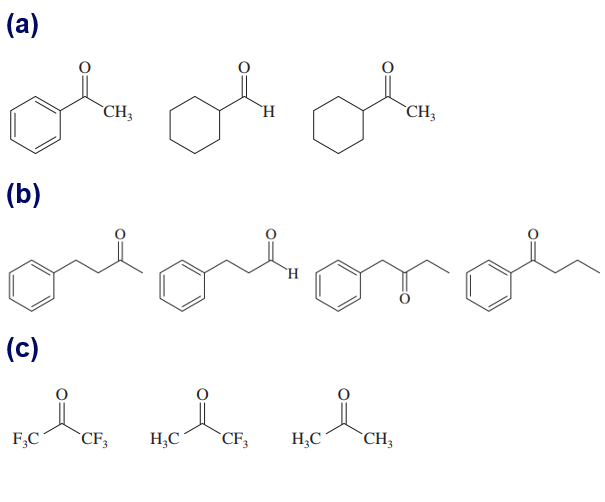
Question: Within each set of structures, indicate which will react fastest, and which slowest, toward nucleophilic addition in basic conditions.
What do you think about this solution?
We value your feedback to improve our textbook solutions.