Chapter 12: Q.19P (page 637)
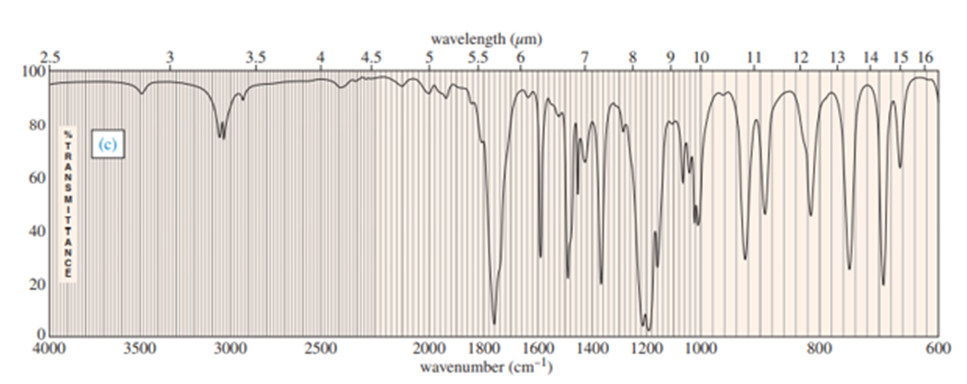
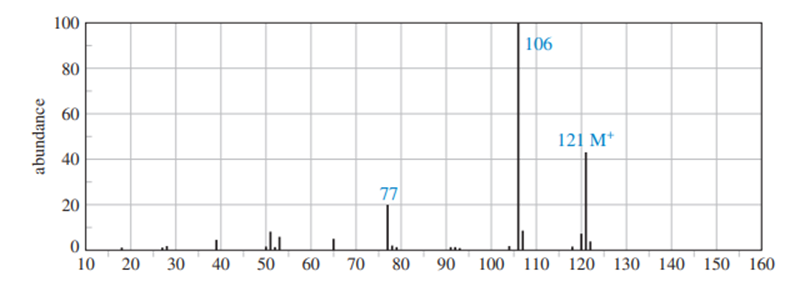
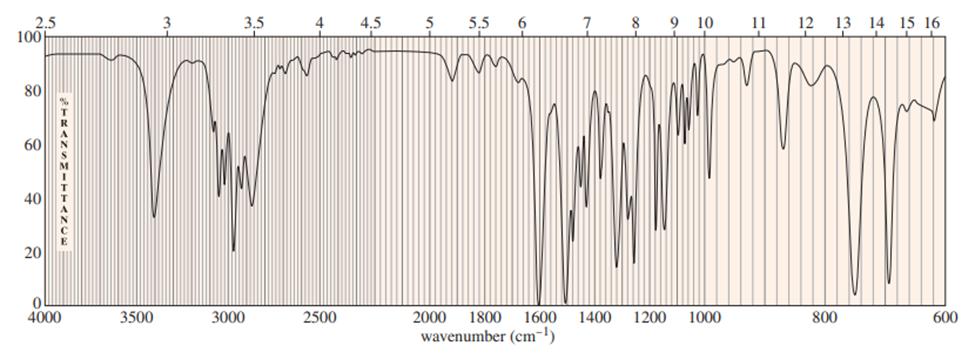
Question: A common lab experiment is the dehydration of cyclopentanol tocyclopentene.
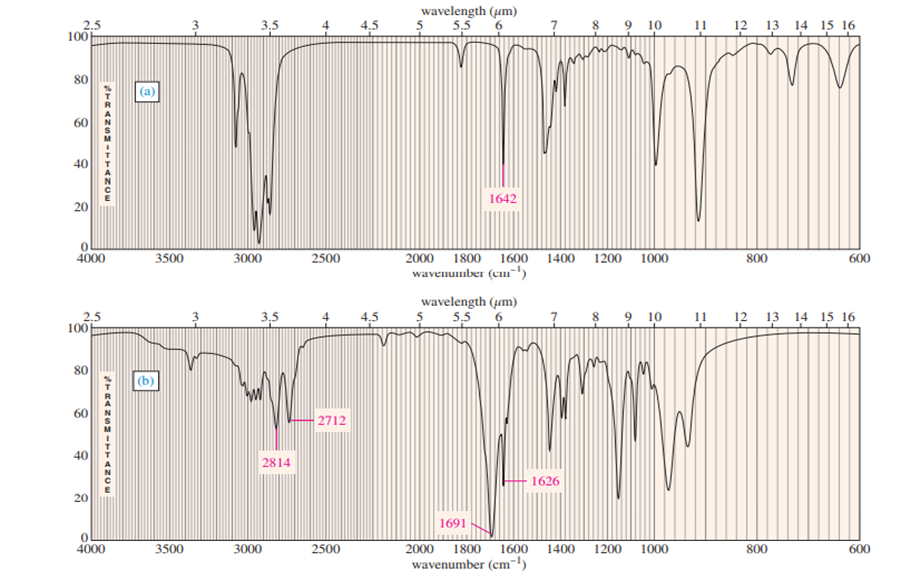
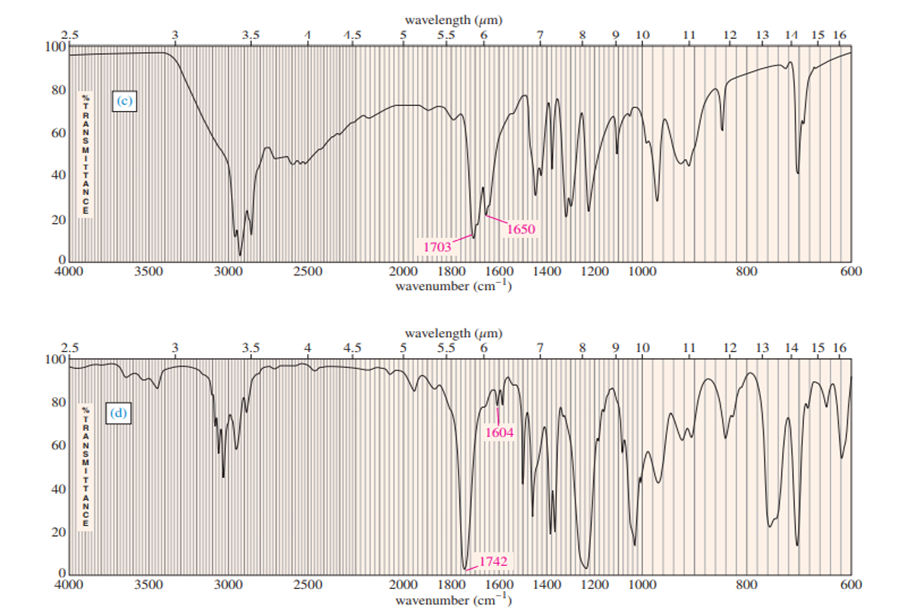
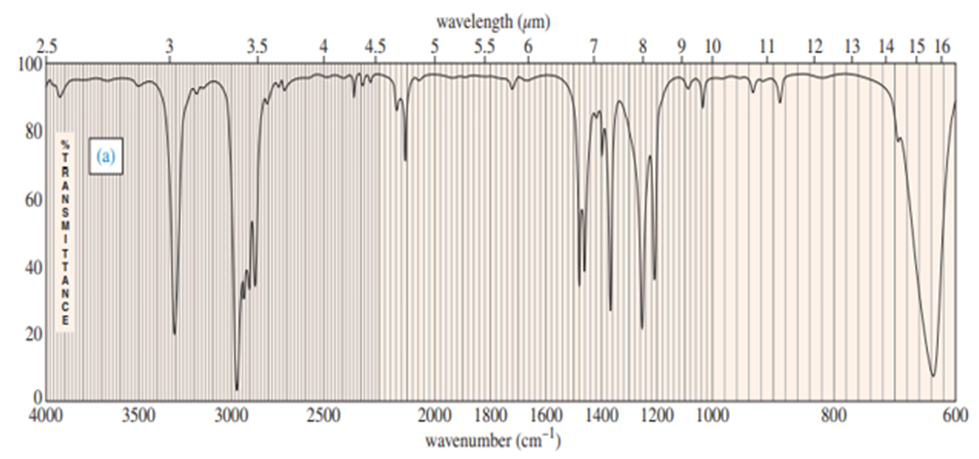
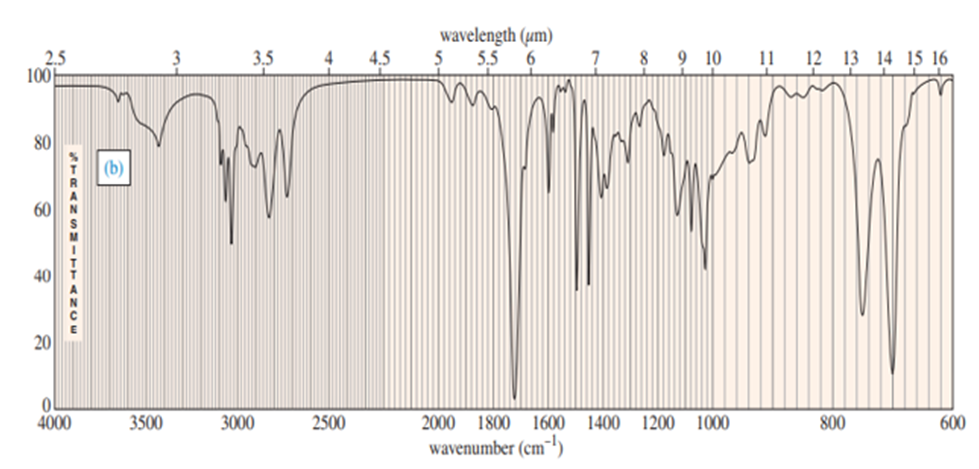
a. Explain how you could tell from the IR spectrum whether your product was pure cyclopentene, pure cyclopentanol or a mixture of cyclopentene and cyclopentanol. Give approximate frequencies for distinctive peaks.
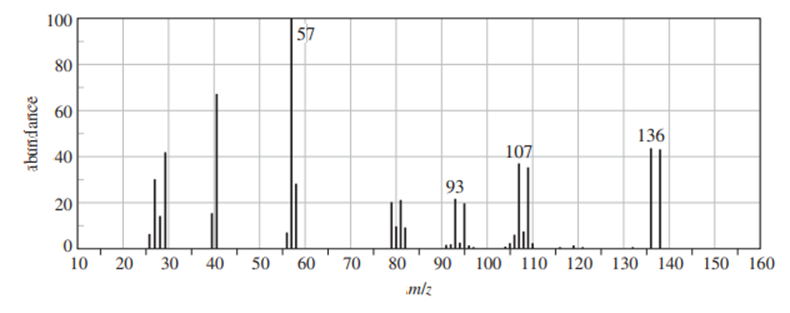
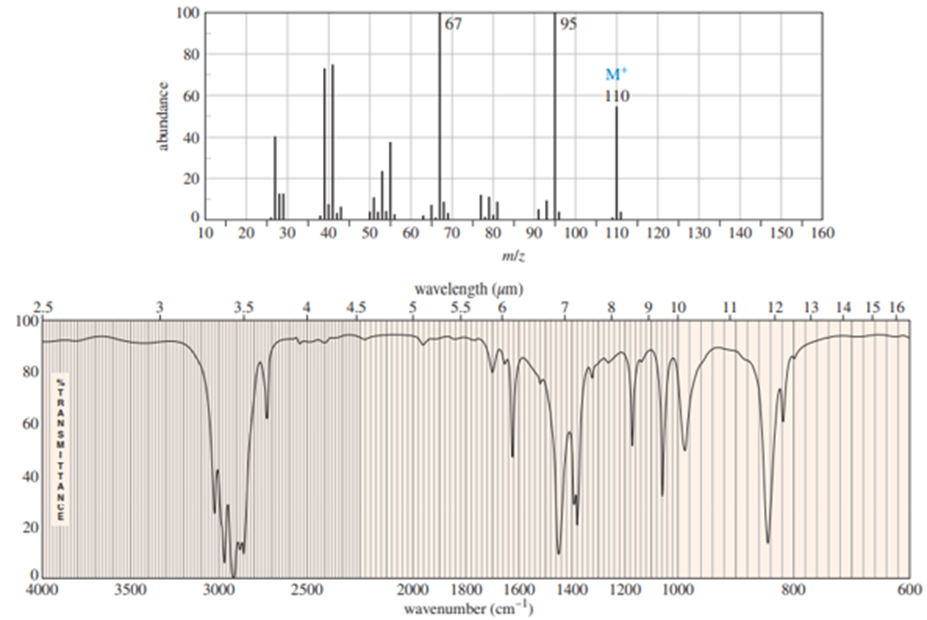
b. Explain why mass spectrometry might not be a good way to distinguish cyclopentene from cyclopentanol.
Short Answer
Answer
a) The compounds can be distinguished on the basis of IR peaks.
b) Mass spectrometry works of the process of fragmentation. Since the compounds are almost identical, mass spectrometry is not a better fit.