Chapter 12: Q25P (page 640)
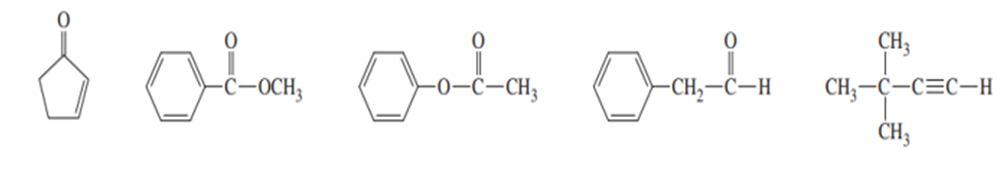
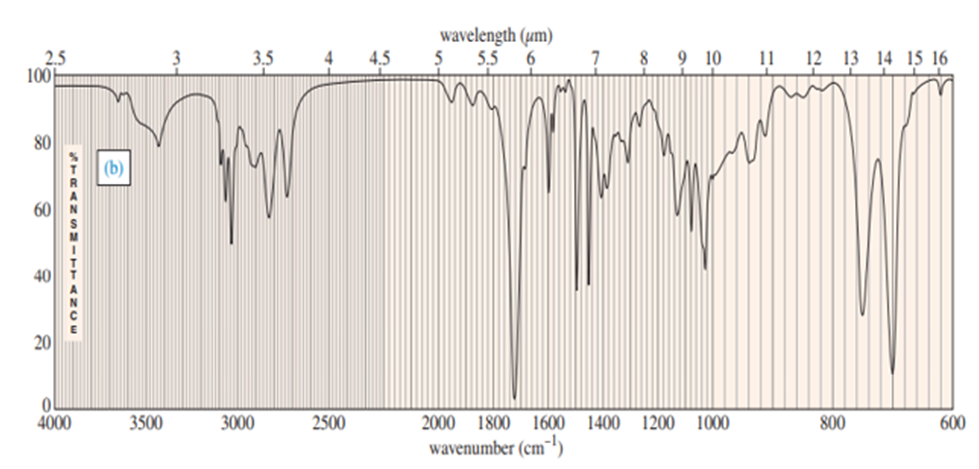
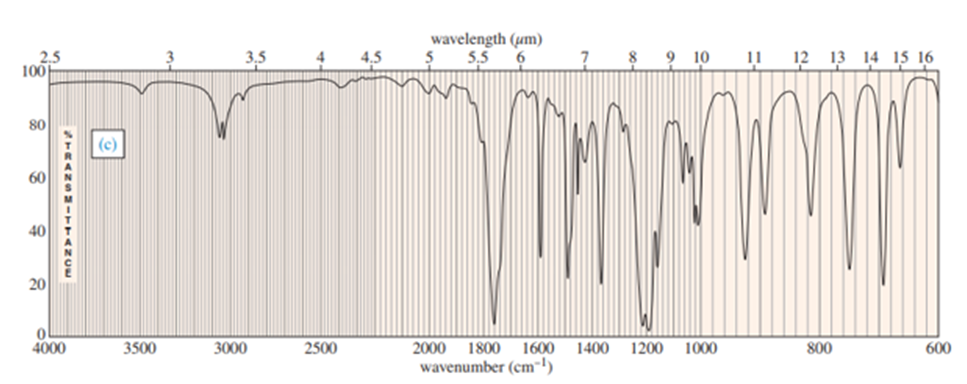
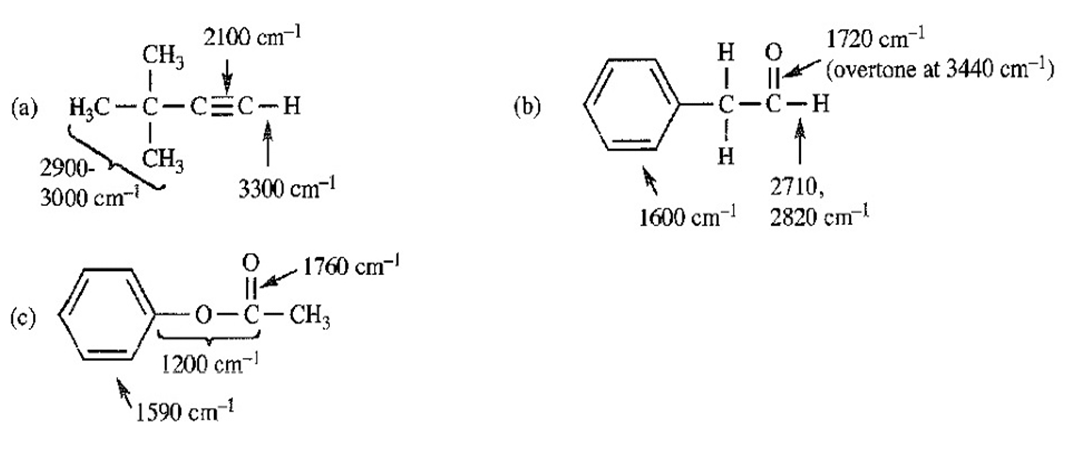
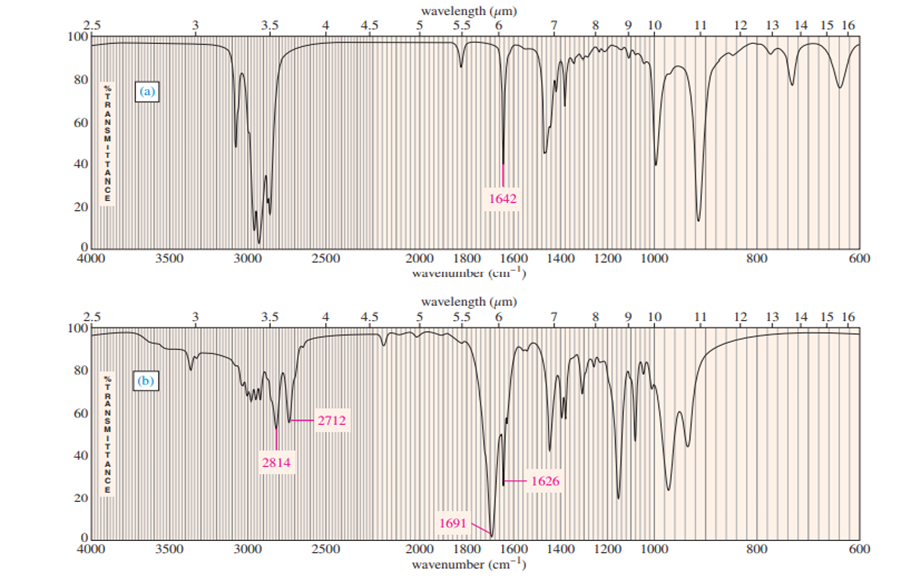
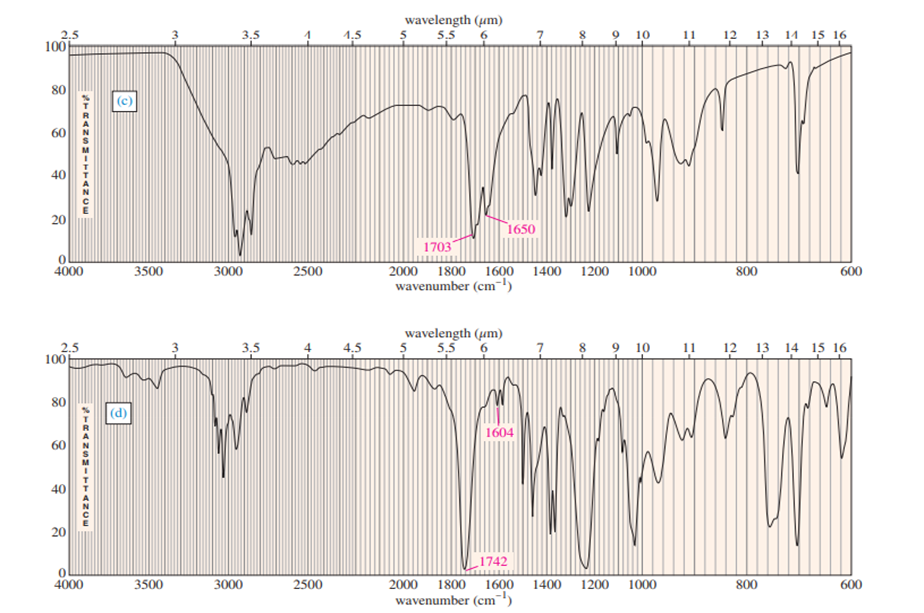
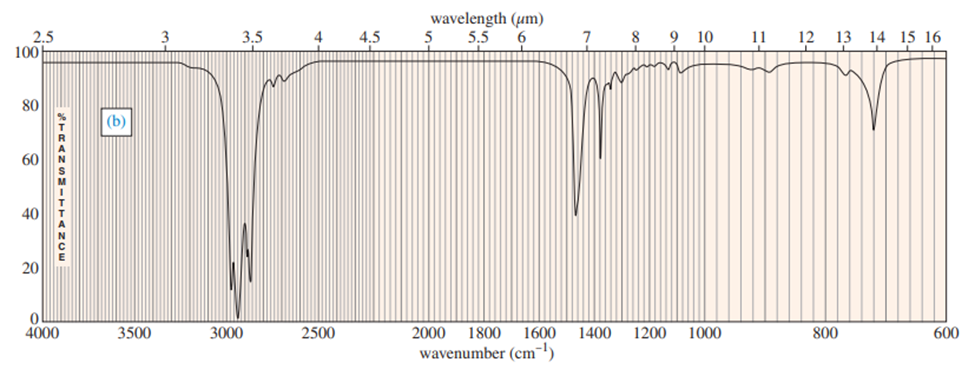
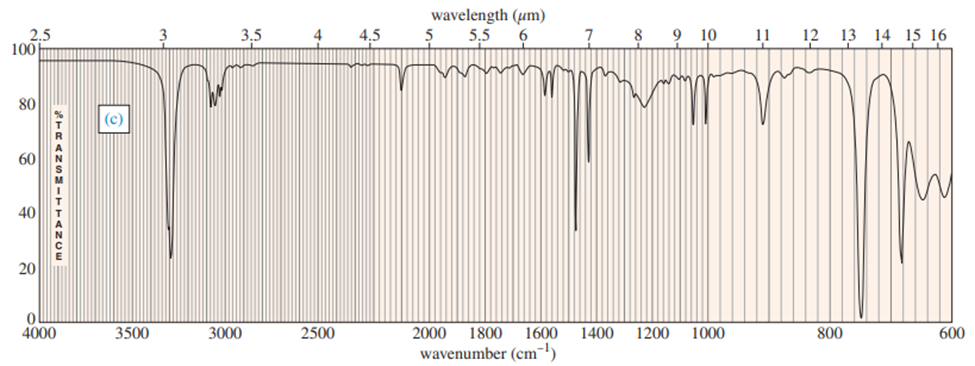
Three IR spectra are shown, corresponding to three of the following compounds. For each spectrum, determine the structure and explain how the peaks in the spectrum correspond to the structure you have chosen.

Short Answer
Learning Materials
Features
Discover
Chapter 12: Q25P (page 640)
Three IR spectra are shown, corresponding to three of the following compounds. For each spectrum, determine the structure and explain how the peaks in the spectrum correspond to the structure you have chosen.





All the tools & learning materials you need for study success - in one app.
Get started for free
The mass spectrum of n-octane shows a prominent molecular ion peak (m/z 114). There is also a large peak at m/z 57, but it is not the base peak. The mass spectrum of 3,4-dimethylhexane shows a smaller molecular ion, and the peak at mass 57 is the base peak. Explain these trends in abundance of the molecular ions and the ions at mass 57 and predict the intensities of the peaks at masses 57 and 114 in the spectrum of 2,2,3,3-tetramethylbutane.
For each spectrum, interpret all the significant streching frequencies above 1580 cm-10
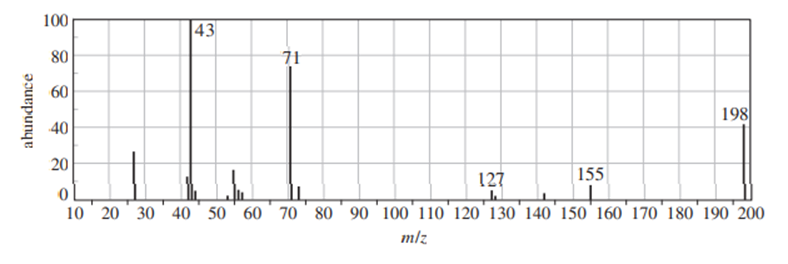
Question: (Another true story.) A student who was checking into her lab desk found an unlabeled sample from a previous student. She was asked to identify the sample. She did an IR spectrum and declared, “It looks like an alkane.” But it seemed too reactive to be an alkane, so she did a GC–MS. The mass spectrum is shown next. Identify the compound as far as you can, and state what part of your identification is uncertain. Propose fragments corresponding to the numbered peaks.
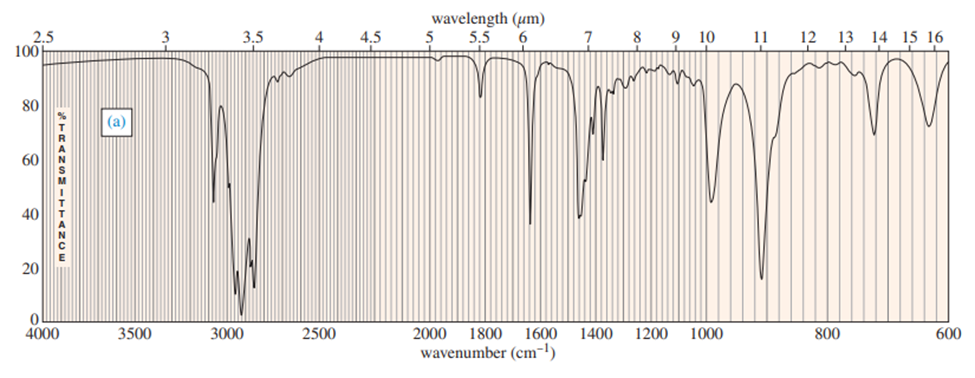
For each hydrocarbon spectrum, determine whether the compound is an alkane, an alkene, an alkyne, or an aromatic hydrocarbon, and assign the major peaks above (to the left of) 1600 cm-1 . More than one unsaturated group may be present.
Chapter-9 covered a synthesis of alkynes by a double dehydrohalogenation of dihalides. A student tried to convert trans-2,5-dimethylhex-3-yne by adding bromine across the double bond and then doing a double elimination. The infrared and mass spectra of the major product are shown here.
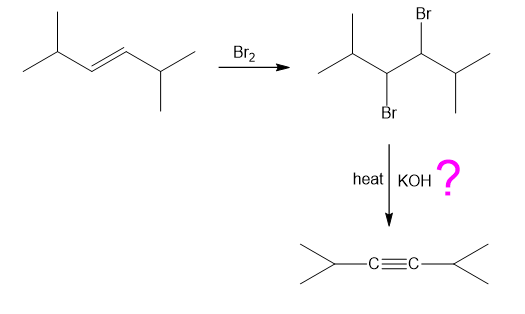

What do you think about this solution?
We value your feedback to improve our textbook solutions.