Chapter 12: Q24P (page 639)
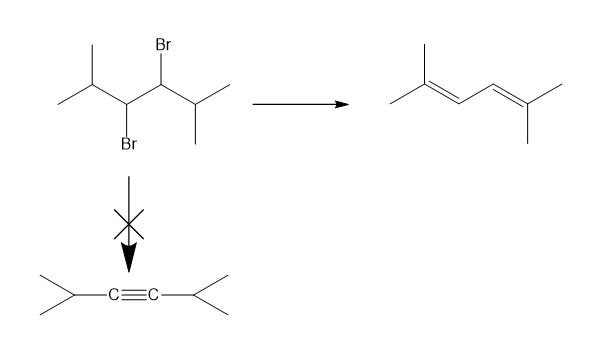
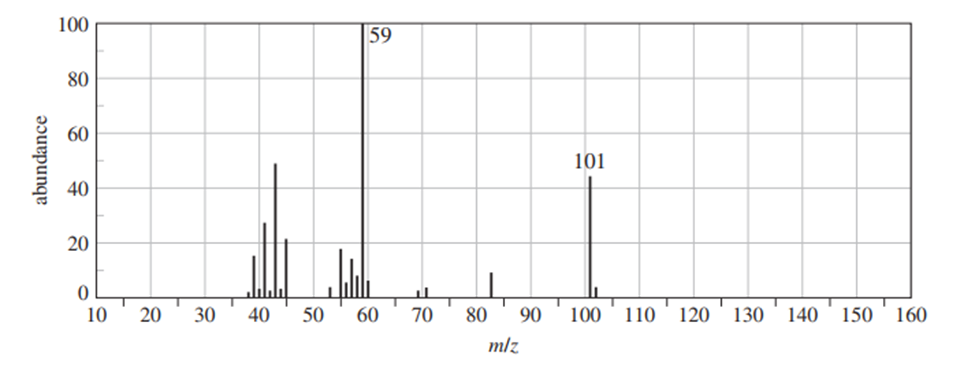
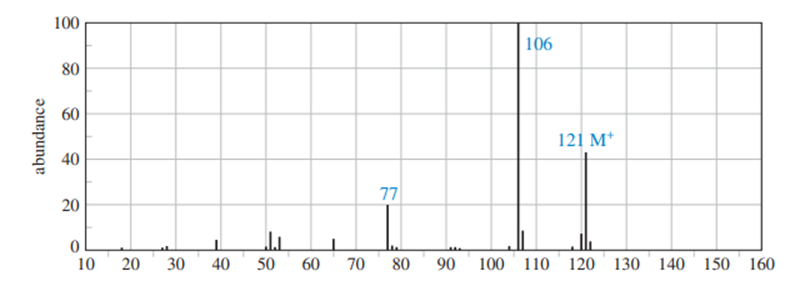
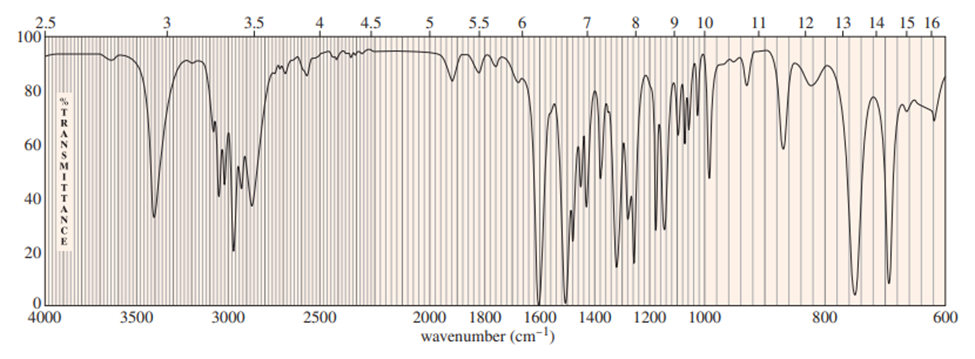
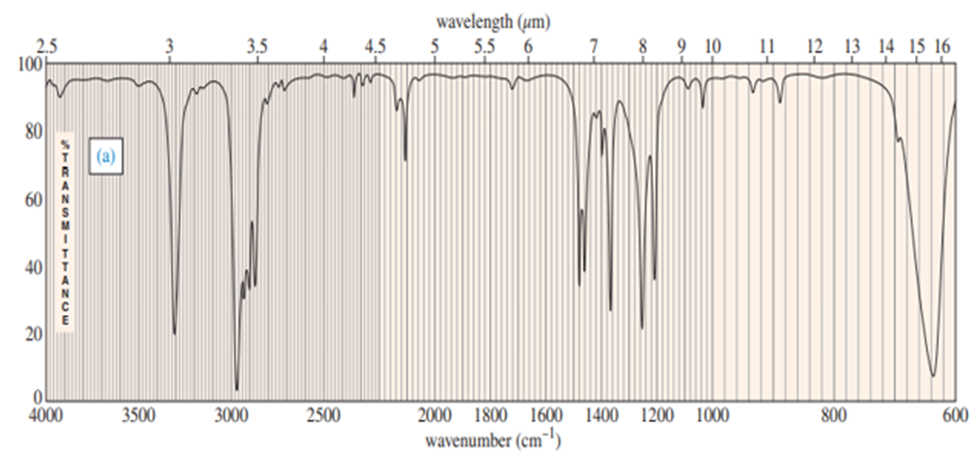
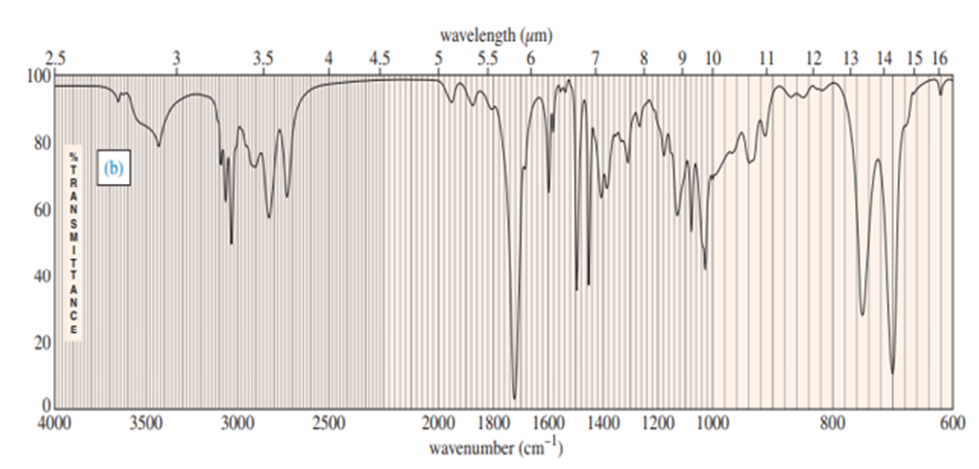
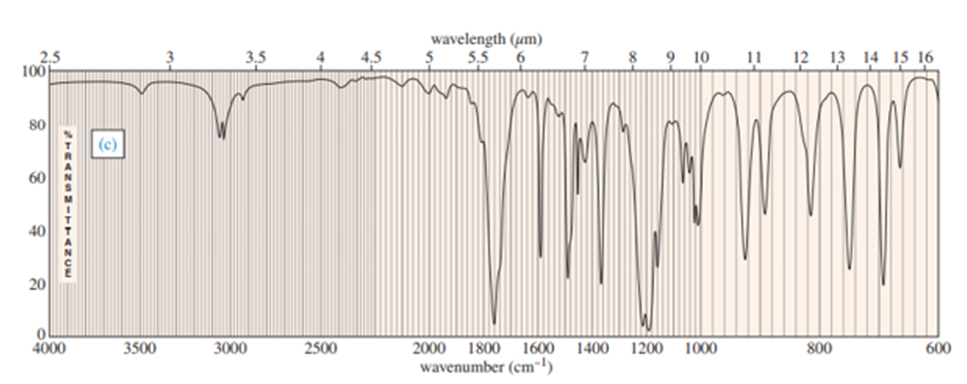
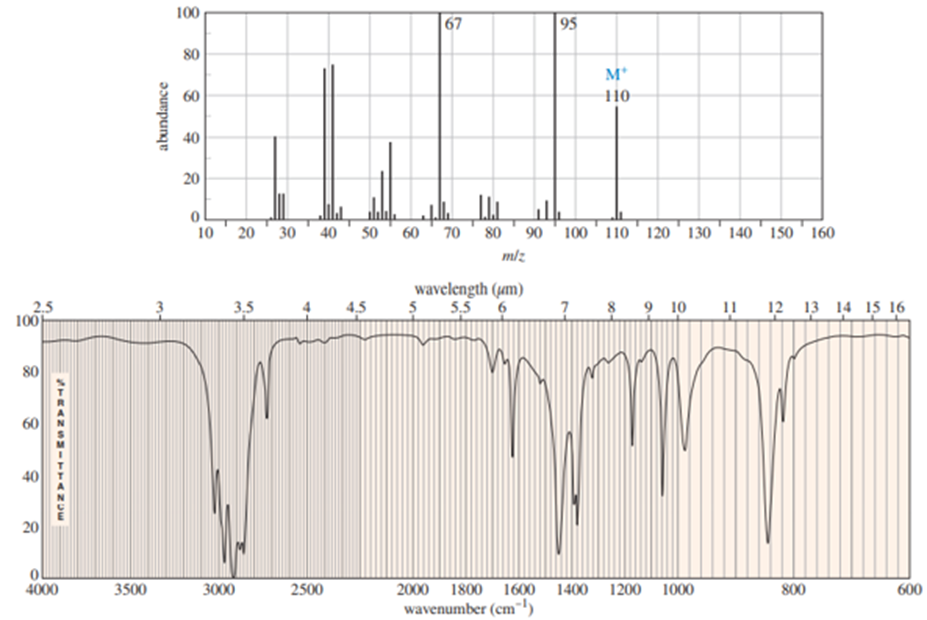
Chapter-9 covered a synthesis of alkynes by a double dehydrohalogenation of dihalides. A student tried to convert trans-2,5-dimethylhex-3-yne by adding bromine across the double bond and then doing a double elimination. The infrared and mass spectra of the major product are shown here.
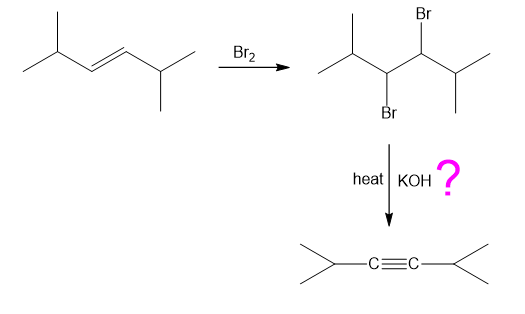
- Do the spectra confirm the right product? If not, what is it?
- Explain the important peaks in the IR spectrum.
Short Answer
The mass spectrum is consistent with the formula of the alkyne m/z 110.The IR is not consistent with the alkyne. Often, symmetrically substituted alkynes have a miniscule peak, so the fact that the IR does not show this peak does not prove that the alkyne is absent.
The important evidence in the IR is the significant peak at 1620 and the absorption above 3000 and this absorption is a characteristic of a conjugated diene. Instead of the alkyne being formed, the reaction must have been a double elimination to the diene.