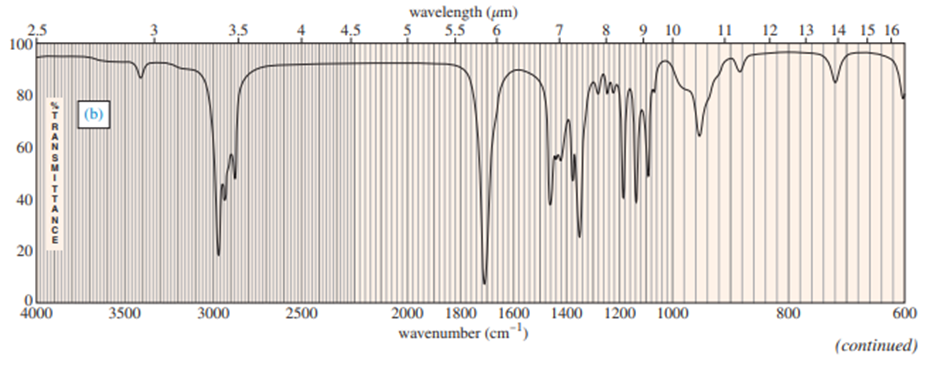
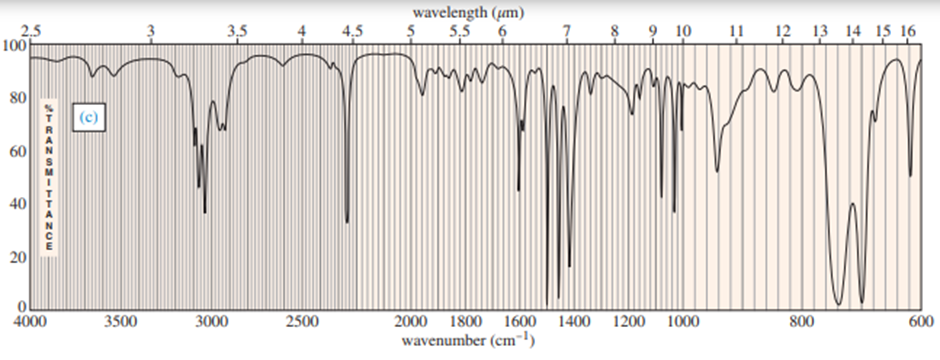
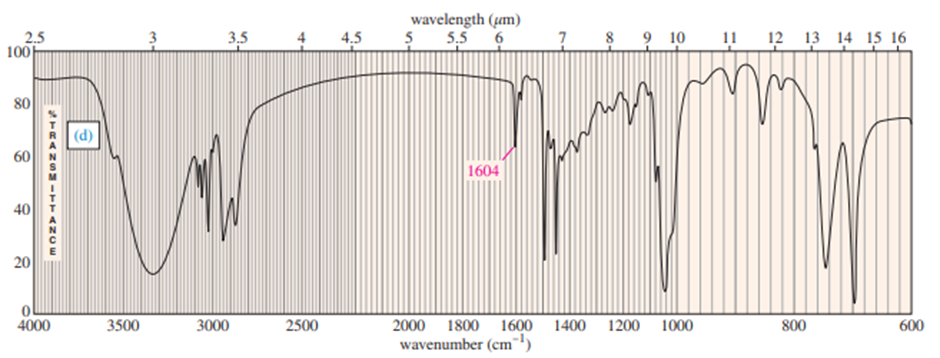
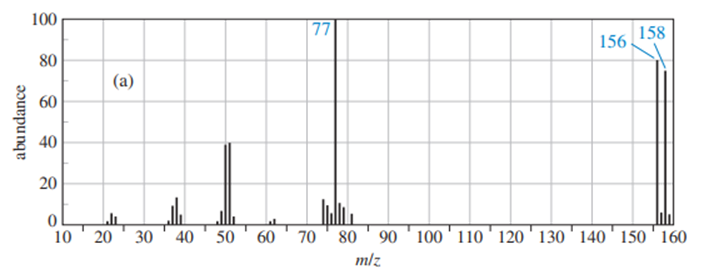
Chapter 12: Q.17P (page 637)
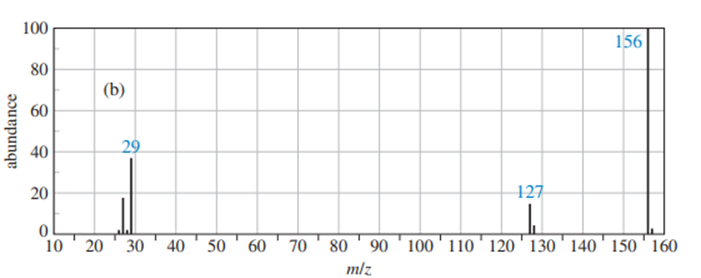
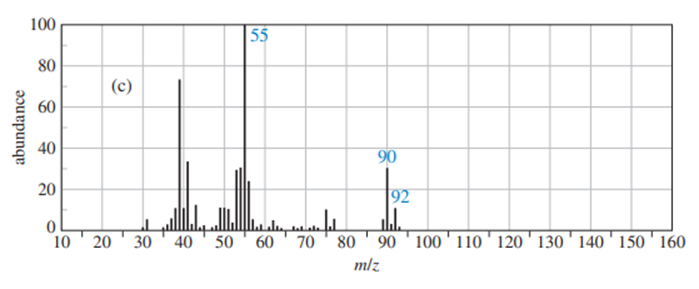
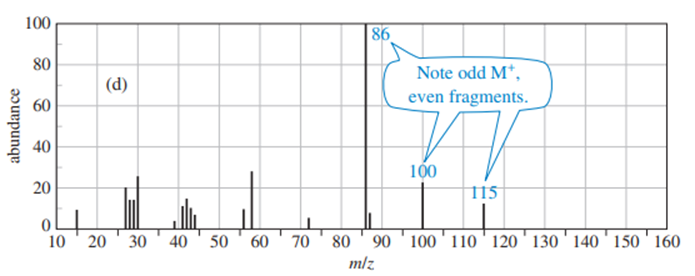
Predict the masses and the structures of the most abundant fragments observed in the mass spectra of the following compounds
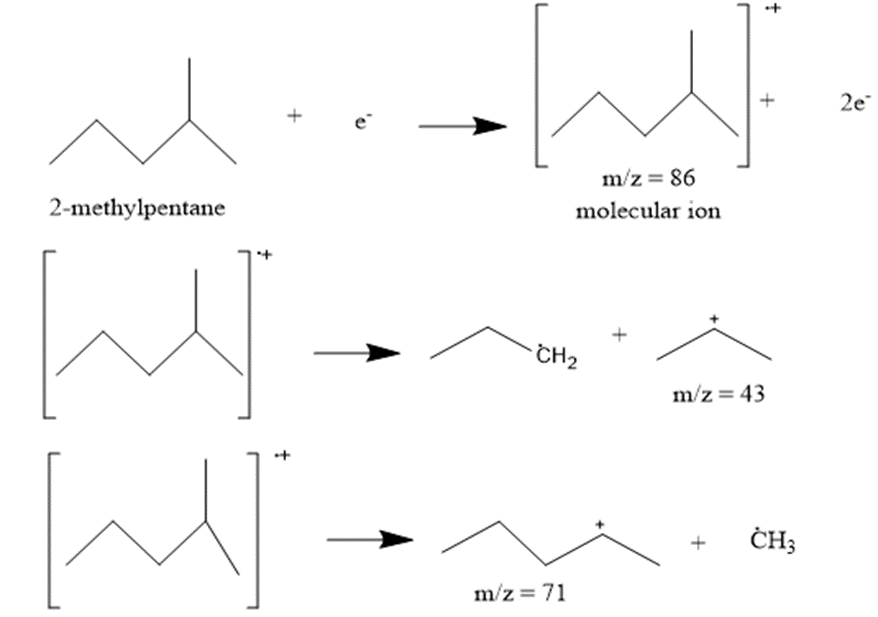
a. 2-methylpentane
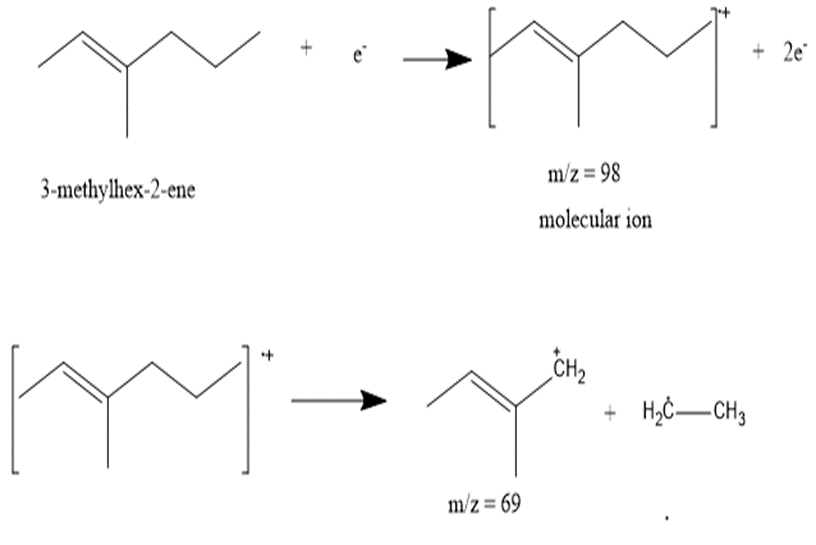
b. 3-methylhex-2-ene
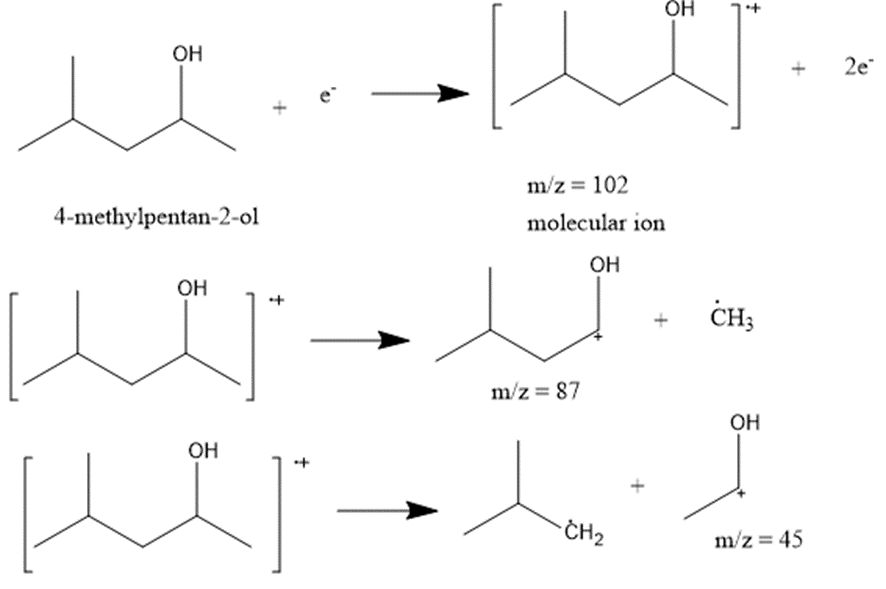
c. 4-methylpentane-2-ol
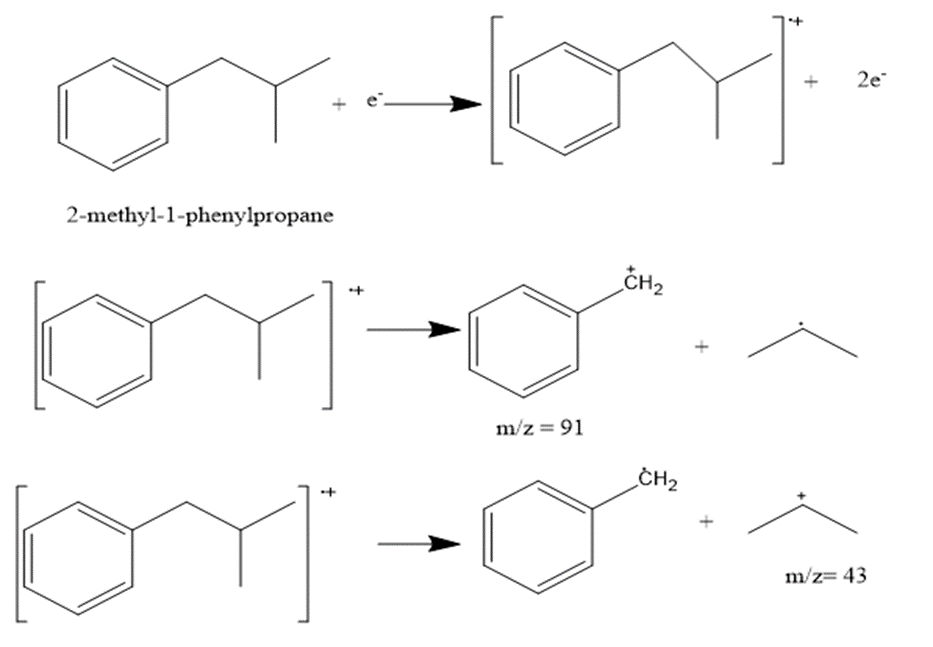
d. 2-methyl-1-phenylpropane
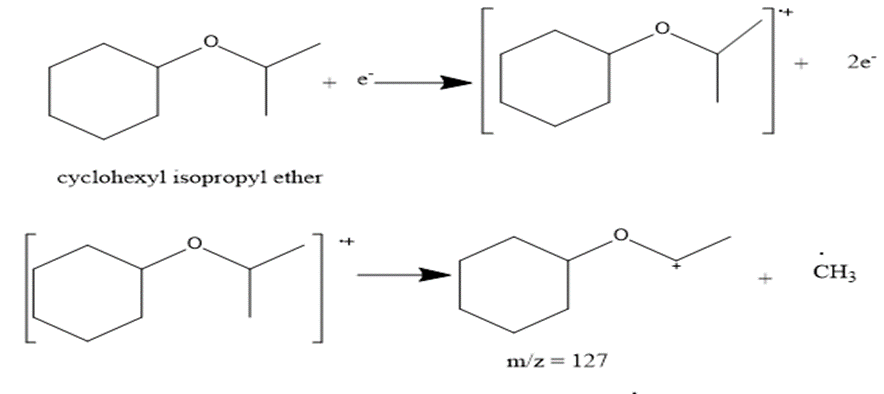
e. Cyclohexylisopropy; ether [cyclohexyl--O-CH(CH3)2 ]
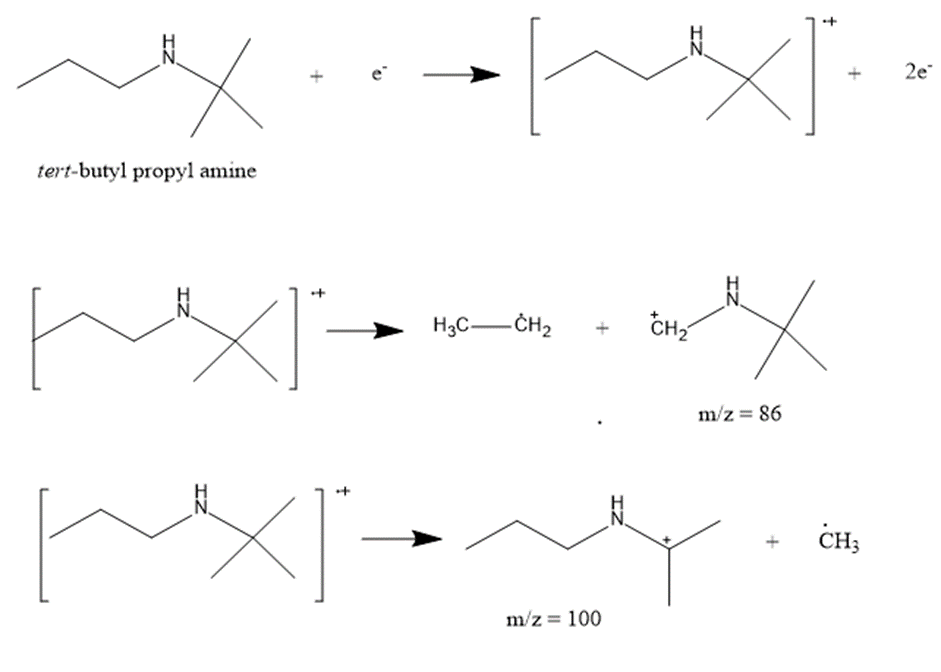
f. CH3CH2CH2NHC(CH3)2
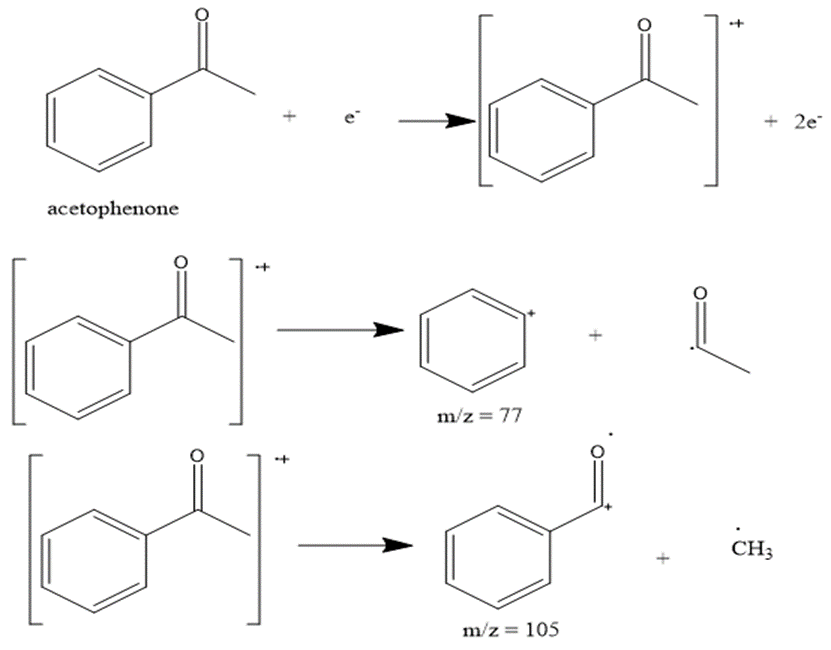
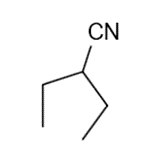
g.
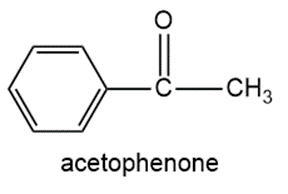
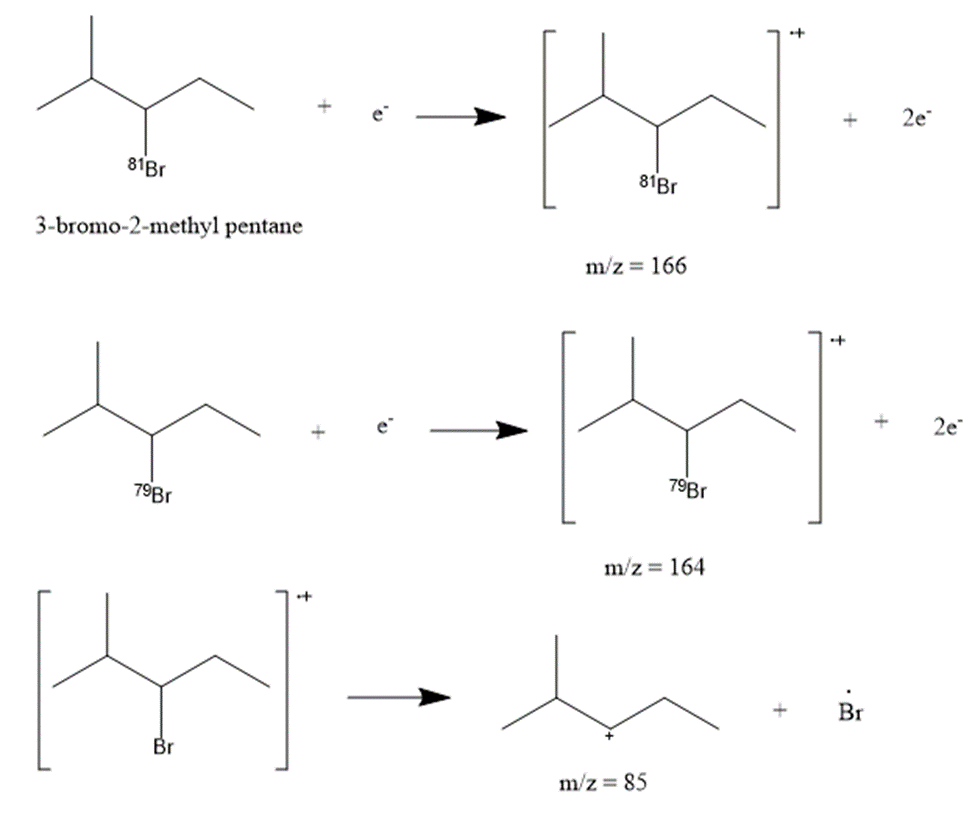
h.3-bromo-2-methylpentane
Short Answer
Answer
The most abundant peaks for all the given species and their formation through fragmentation is shown.