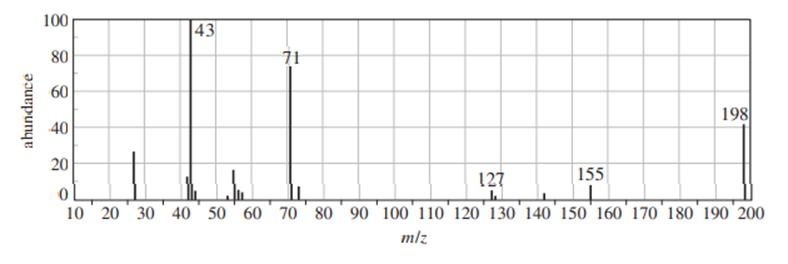
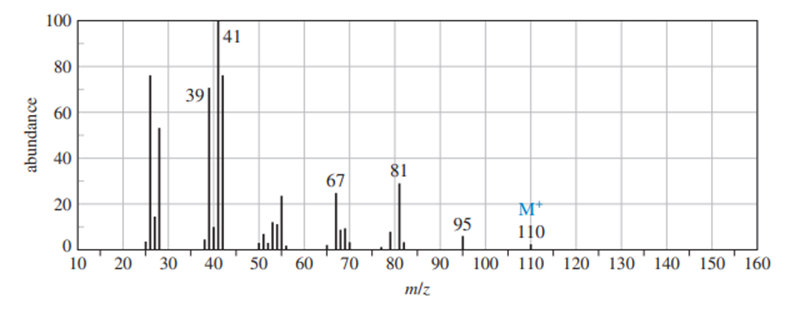
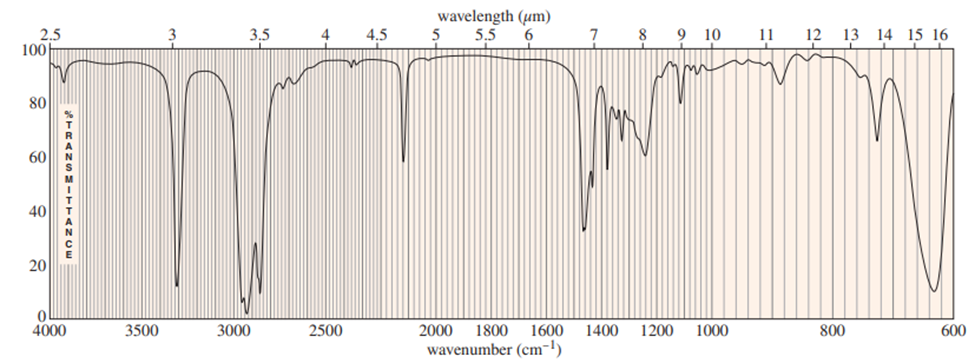
Chapter 12: Q16P (page 625)
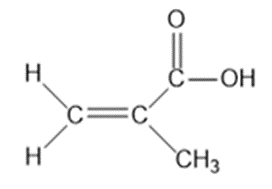
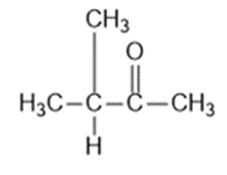
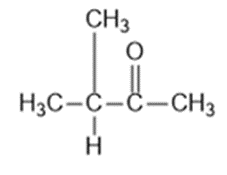
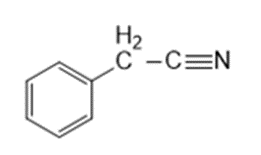
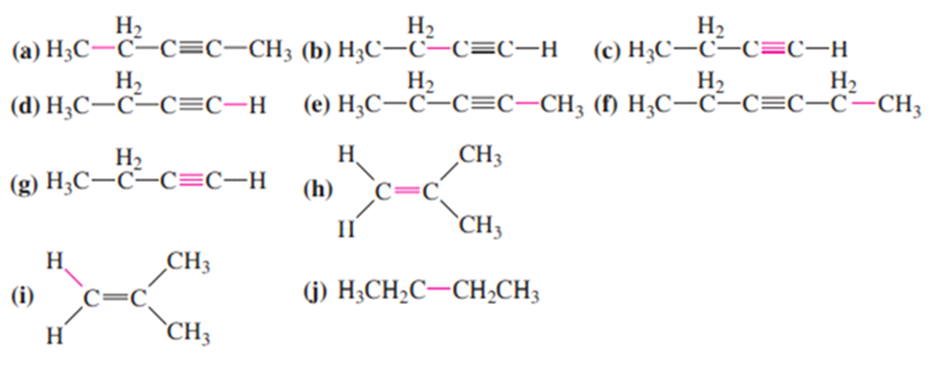
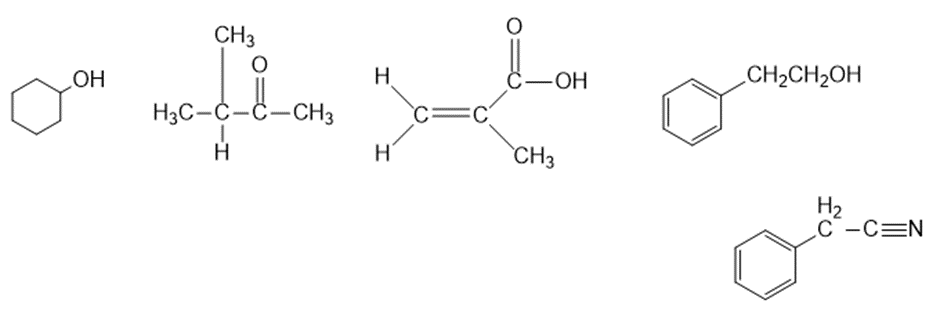
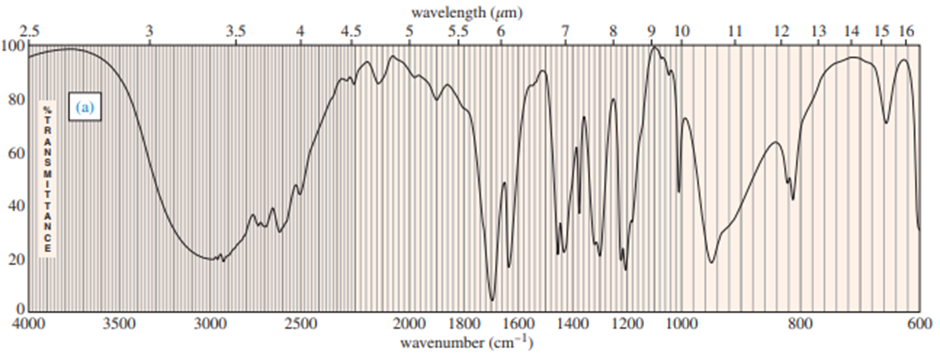
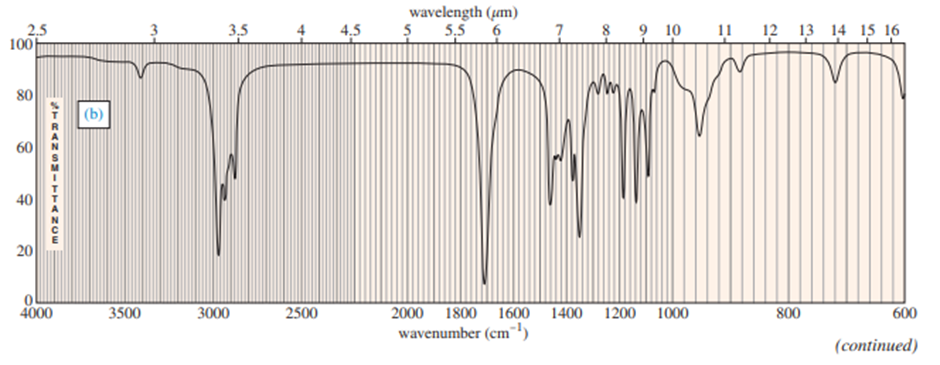
Question: Four infrared spectra are shown, corresponding to four of the following compounds. For each spectrum, determine the structure and explain how the peaks in the spectrum correspond to the structure you have chosen
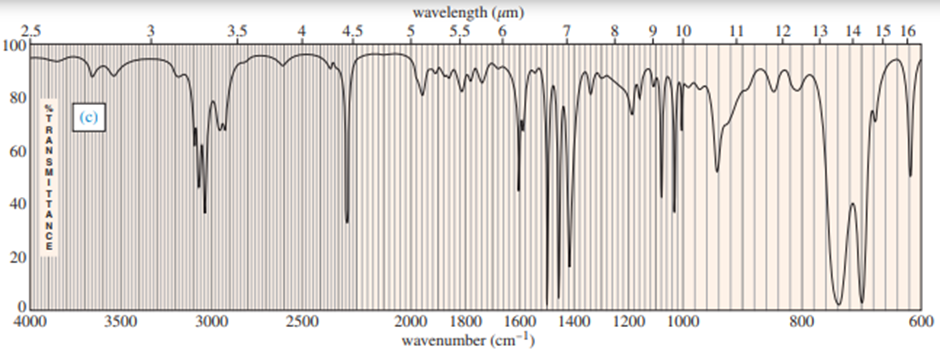
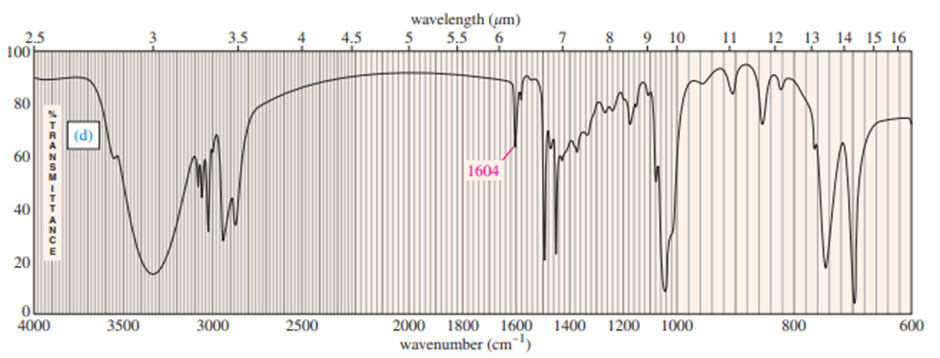
Short Answer
Answer