Chapter 12: Q1P (page 595)
Complete the following conversion table:
4000 | 1800 | 1670 | 1620 | 400 | ||||
2.50 | 3.40 | 3.13 | 4.87 | 25.0 |
Short Answer
4000 | 2941.17 | 3095.97 | 2053.38 | 1800 | 1670 | 1620 | 400 | |
2.50 | 3.40 | 3.13 | 4.87 | 5.55 | 5.98 | 6.17 | 25.0 |
Learning Materials
Features
Discover
Chapter 12: Q1P (page 595)
Complete the following conversion table:
4000 | 1800 | 1670 | 1620 | 400 | ||||
2.50 | 3.40 | 3.13 | 4.87 | 25.0 |
4000 | 2941.17 | 3095.97 | 2053.38 | 1800 | 1670 | 1620 | 400 | |
2.50 | 3.40 | 3.13 | 4.87 | 5.55 | 5.98 | 6.17 | 25.0 |
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: (A true story.) While organizing the undergraduate stockroom, a new chemistry professor found a half-gallon jug containing a cloudy liquid (bp 100–105 °C), marked only “STUDENT PREP.” She ran a quick mass spectrum, which is printed below. As soon as she saw the spectrum (without even checking the actual mass numbers), she said, “I know what it is.”

(a) What compound is the “student prep”? Any uncertainty in the structure?
(b) Suggest structures for the fragments at 136, 107, and 93. Why is the base peak (at m/z 57) so strong?
The mass spectrum of n-octane shows a prominent molecular ion peak (m/z 114). There is also a large peak at m/z 57, but it is not the base peak. The mass spectrum of 3,4-dimethylhexane shows a smaller molecular ion, and the peak at mass 57 is the base peak. Explain these trends in abundance of the molecular ions and the ions at mass 57 and predict the intensities of the peaks at masses 57 and 114 in the spectrum of 2,2,3,3-tetramethylbutane.
These five structures all have distinguishing absorption in the IR. Match each structure with its characteristic absorption.
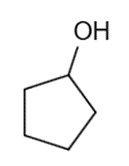
Structure 1
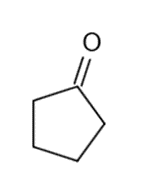
Structure 2

Structure 3
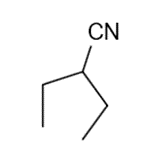
Structure 4
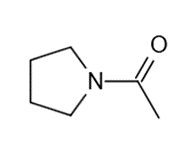
Structure 5
(a)sharp, 2254 cm-1 (b) very broad, centered about3330cm-1 (c) strong, slightly broadened, 1645cm-1 (d) broad with spikes at 3367cm-1and 3392cm-1(e) strong, sharp 1717cm-1
Question: Show the fragmentation that accounts for the cation at m/z 57 in the mass spectrum of 2-methylpentane. Explain why this ion is less abundant than those at m/z 71 and 43.
Question: Consider the following four structures, followed by mass spectral data. Match each structure with its characteristic molecular ion or fragment. In each case, give a likely structure of the ion responsible for the base peak

(a) base peak at 105
(b) base peak at 72
(c) doublet at 198 and 200, base peak at 91
(d) base peak at 91, large peak at 43
What do you think about this solution?
We value your feedback to improve our textbook solutions.