Chapter 12: Q.8P (page 629)
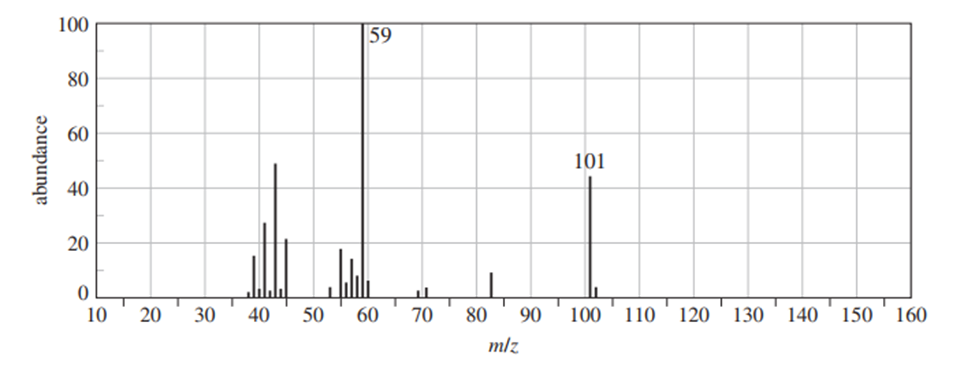
Question: Show the fragmentation that accounts for the cation at m/z 57 in the mass spectrum of 2-methylpentane. Explain why this ion is less abundant than those at m/z 71 and 43.
Short Answer
Answer
Abundance of ion depends upon the stability of ion as the stability order of ions (Carbonations) M/z=43>M/z=71>M/z=57
as M/z =43 ion 2o carbocation with 6 α-hydrogens so, the most stable followed by M/z=71 carbocation M/z=71 1o carbocation with 2 α-hydrogens and least stable M/z=57 ion 1o carbocation having only 1-α hydrogen.