Chapter 12: Q.9P (page 629)
Show the fragmentations that give rise to the peaks at m/z 43, 57, and 85 in the mass spectrum of 2,4-dimethylpentane (Figure 12-17).
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 12: Q.9P (page 629)
Show the fragmentations that give rise to the peaks at m/z 43, 57, and 85 in the mass spectrum of 2,4-dimethylpentane (Figure 12-17).
Answer
All the tools & learning materials you need for study success - in one app.
Get started for free
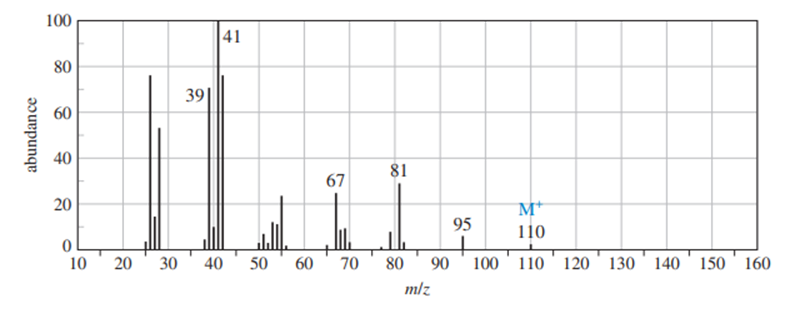
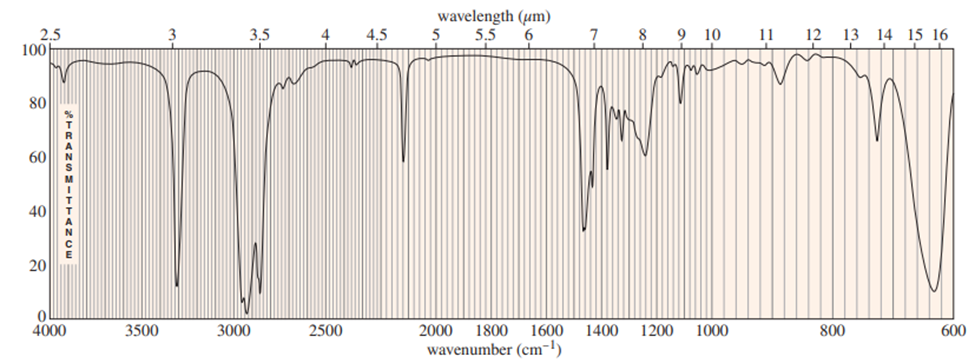
Question: (a) Use the mass spectrum to propose a molecular formula. How many elements of unsaturation are there? (b) Use the IR spectrum to determine the functional group(s), if any. (c) Propose one or more structures for this compound. What parts of the structure are uncertain? If you knew that hydrogenation of the compound gives n-octane, would the structure still be uncertain? (d) Propose structures for the major fragments at 39, 67, 81, and 95 in the mass spectrum.
These five structures all have distinguishing absorption in the IR. Match each structure with its characteristic absorption.
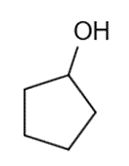
Structure 1
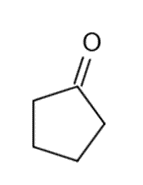
Structure 2
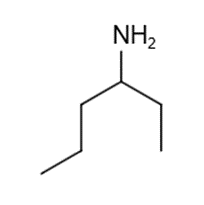
Structure 3
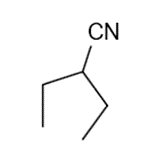
Structure 4
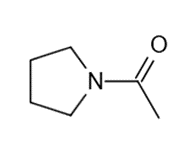
Structure 5
(a)sharp, 2254 cm-1 (b) very broad, centered about3330cm-1 (c) strong, slightly broadened, 1645cm-1 (d) broad with spikes at 3367cm-1and 3392cm-1(e) strong, sharp 1717cm-1
Question: Show the fragmentation that accounts for the cation at m/z 57 in the mass spectrum of 2-methylpentane. Explain why this ion is less abundant than those at m/z 71 and 43.
Question:Spectra are given for three compounds. Each compound has one or more of the following functional groups: alcohol, amine, ketone, aldehyde, and carboxylic acid. Determine the functional group(s) in each compound, and assign the major peaks above 1600cm-1.
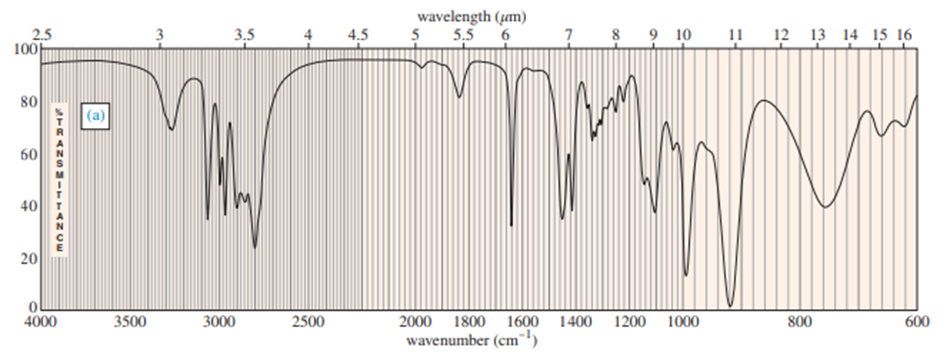
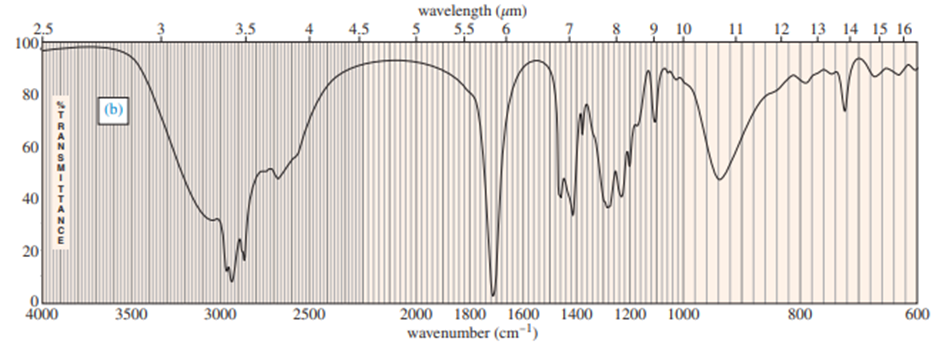
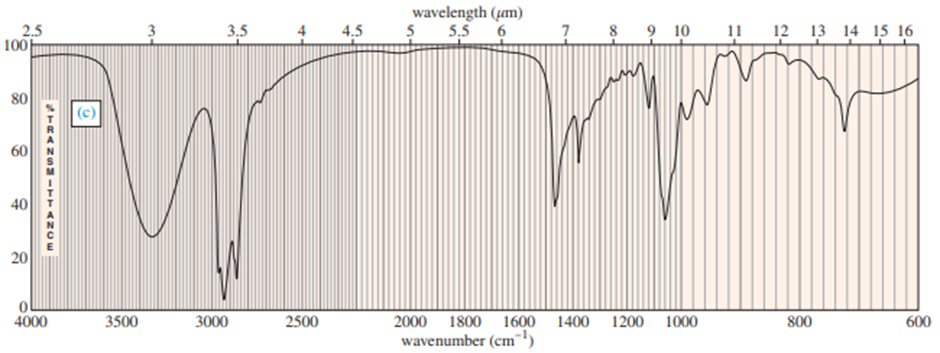
Question: (A true story.) While organizing the undergraduate stockroom, a new chemistry professor found a half-gallon jug containing a cloudy liquid (bp 100–105 °C), marked only “STUDENT PREP.” She ran a quick mass spectrum, which is printed below. As soon as she saw the spectrum (without even checking the actual mass numbers), she said, “I know what it is.”
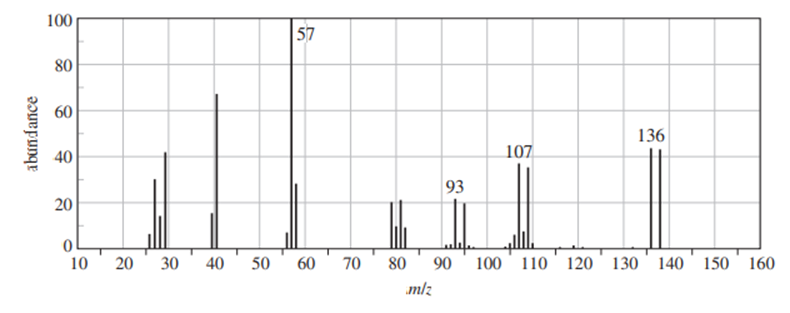
(a) What compound is the “student prep”? Any uncertainty in the structure?
(b) Suggest structures for the fragments at 136, 107, and 93. Why is the base peak (at m/z 57) so strong?
What do you think about this solution?
We value your feedback to improve our textbook solutions.