H. G. Khorana won the Nobel Prize in Medicine in 1968 for developing the synthesis of DNA and RNA and for helping to unravel the genetic code. Part of the chemistry he developed was the use of selective protecting groups for the 5′ OH group of nucleosides.
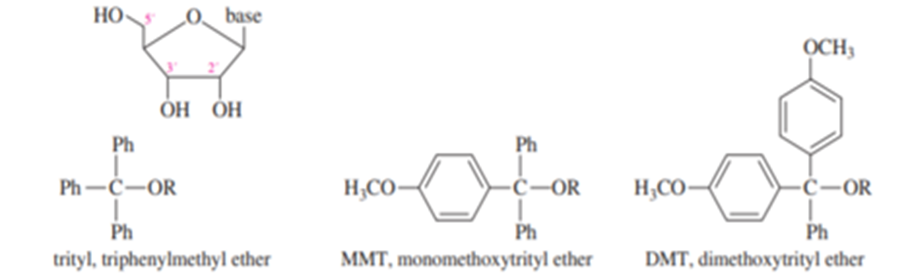
The trityl ether derivative of just the 5′ OH group is obtained by reaction of the nucleoside with trityl chloride, MMT chloride, or DMT chloride and a base like Et3N. The trityl ether derivative can be removed in dilute aqueous acid. DMT derivatives hydrolyze fastest, followed by MMT derivatives, and trityl derivatives slowest.
(a) Draw the product with the trityl derivative on the 5′ oxygen.
(b) Explain why the trityl derivative is selective for the 5′ OH group. Why doesn’t it react at 2′ or 3′? (c) Why is the DMT group easiest to remove under dilute acid conditions? Why does the solution instantly turn orange when acid is added to a DMT derivative?





