Chapter 23: Q21P (page 1225)
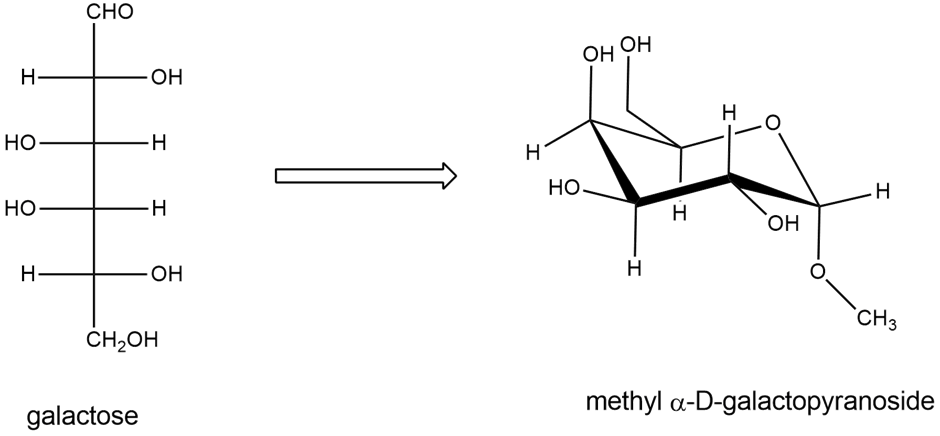
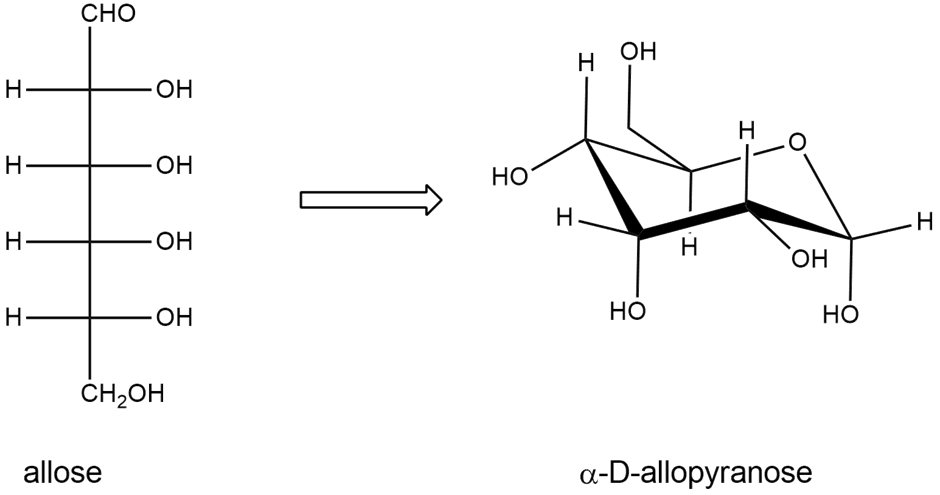
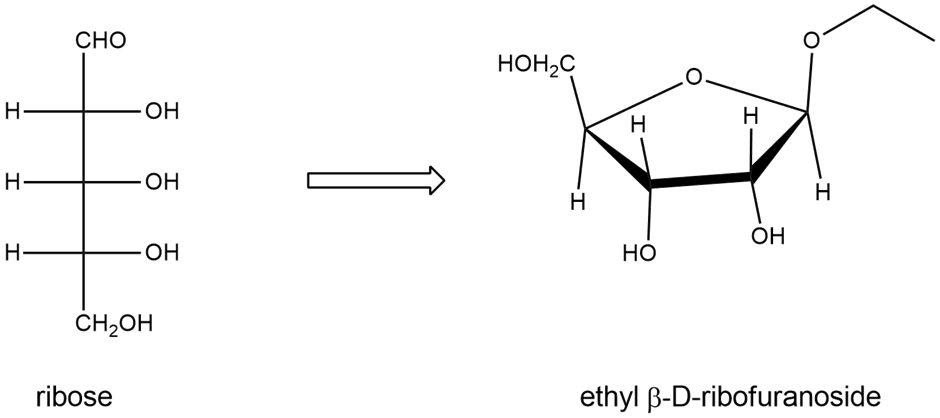
Draw the structures of the compounds named in Problem 23-20 parts (a), (c), and (d). Allose is the C3 epimer of glucose and ribose is the C2 epimer of arabinose.
Short Answer
Learning Materials
Features
Discover
Chapter 23: Q21P (page 1225)
Draw the structures of the compounds named in Problem 23-20 parts (a), (c), and (d). Allose is the C3 epimer of glucose and ribose is the C2 epimer of arabinose.



All the tools & learning materials you need for study success - in one app.
Get started for free
The relative configurations of the stereoisomers of tartaric acid were established by the following synthesis:
(1) D-(+)-glyceraldehyde
(2) Hydrolysis of A and B using aqueous Ba(OH)2 gave C and D, respectively.
(3) HNO3 oxidation of C and D gave (-)-tartaric acid and meso-tartaric acid, respectively.
(a) You know the absolute configuration of D-(+)-glyceraldehyde, Use Fischer projections to show the absolute configurations of products A, B, C, and D.
(b) Show the absolute configurations of the three stereoisomers of tartaric acid: (+)-tartaric acid, (-)-tartaric acid, and meso-tartaric acid.
Except for the Tollens test, basic aqueous conditions are generally avoided with sugars because they lead to fast isomerizations.
Predict the products formed when the following sugars react with excess acetic anhydride and pyridine.
Question:
a) Draw D-allose, the C3 epimer of glucose.
b) Draw D-talose, the C2 epimer of D-galactose.
c) Draw D-idose, the C3 epimer of D-talose. Now compare your answers with Figure 23-3.
d) Draw the C4 “epimer” of D-xylose. Notice that this “epimer” is actually an L-series sugar, and we have seen its enantiomer. Give the correct name for this L-series sugar.
Emil Fischer synthesized L-gulose, an unusual aldohexose that reduces to give D-glucitol. Suggest a structure for this L sugar, and show how L-gulose gives the same alditol as D-glucose. (Hint: D-Glucitol has -CH2OHgroups at both ends. Either of these primary alcohol groups might have come from reduction of an aldehyde.)
What do you think about this solution?
We value your feedback to improve our textbook solutions.