Chapter 23: Q20. (page 1225)
Which of the following sugars are reducing sugars? Comment on the common name sucrose for table sugar.
(a) (b) (an aldohexose)
(c) (d)
(e) (f)

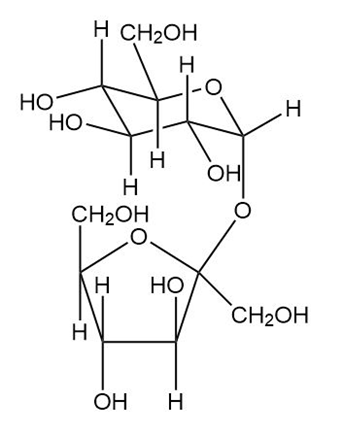
Short Answer
(a) non-reducing sugar
(b) reducing sugar
(c) reducing sugar
(d) non-reducing sugar
(e) reducing sugar
(f) non-reducing sugar



