Chapter 23: Question 53 (page 1255)
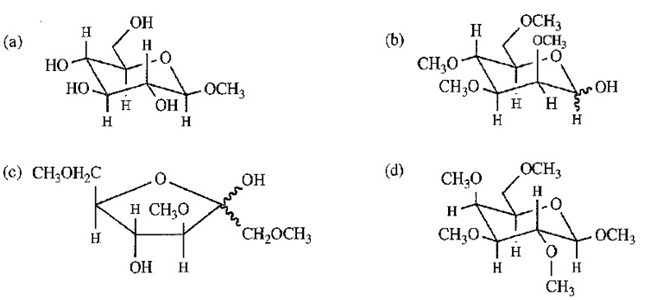
Draw the following sugar derivatives.
- ethyl--D-glucopyranoside
- 2,3,4,6-tetra-O-ethyl-D-mannopyranose
- 1,3,6-tri-O-ethyl-D-fructofuranose
- Ethyl-2,3,4,6-tetra-O-methyl--D-galactopyranoside
Short Answer
Answer :
Learning Materials
Features
Discover
Chapter 23: Question 53 (page 1255)
Draw the following sugar derivatives.
Answer :

All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the structures (using chair conformations of pyranoses) of the following disaccharides.
(a) 4-O-(α -D-glucopyranosyl)-D-galactopyranose
(b) α -D-fructofuranosyl-β -D-mannopyranoside
(c) 6-O-(β -D-galactopyranosyl)-D-glucopyranose
Question. Retroviruses like HIV, the pathogen responsible for AIDS, incorporate an RNA template that is copied into DNA during infection. The reverse transcriptaseenzyme that copies RNA into DNA is relatively nonselective and error-prone, leading to a high mutation rate. Its lack of selectivity is exploited by the anti-HIV drug AZT (3’-azido-2’,3’-dideoxythymidine), which becomes phosphorylated and is incorporated by reverse transcriptase into DNA, where it acts as a chain terminator. Mammalian DNA polymerases are more selective, having a low affinity for AZT, so its toxicity is relatively low.
Erwin Chargaff’s discovery that DNA contains equimolar amounts of guanine and cytosine and also equimolar amounts of adenine and thymine has come to be known as Chargaff’s rule:
G = C and A = T
(a) Does Chargaff’s rule imply that equal amounts of guanine and adenine are present in DNA? That is, does G = A?
(b) Does Chargaff’s rule imply that the sum of the purine residues equals the sum of the pyrimidine residues? That is, does A + G = C + T?
(c) Does Chargaff’s rule apply only to double-stranded DNA, or would it also apply to each individual strand if the double helical strand were separated into its two complementary strands?
Predict the products formed when the following sugars react with excess acetic anhydride and pyridine.
Give an equation to show the reduction of Tollens reagent by maltose.
What do you think about this solution?
We value your feedback to improve our textbook solutions.