Chapter 23: Q37P (page 1236)
Give an equation to show the reduction of Tollens reagent by maltose.
Short Answer
The reduction of Tollens reagent by maltose is shown below.
Learning Materials
Features
Discover
Chapter 23: Q37P (page 1236)
Give an equation to show the reduction of Tollens reagent by maltose.
The reduction of Tollens reagent by maltose is shown below.
All the tools & learning materials you need for study success - in one app.
Get started for free
. (a) An aliphatic aminoglycoside is relatively stable to base, but it is quickly hydrolyzed by dilute acid. Propose a mechanism for the acid-catalyzed hydrolysis.
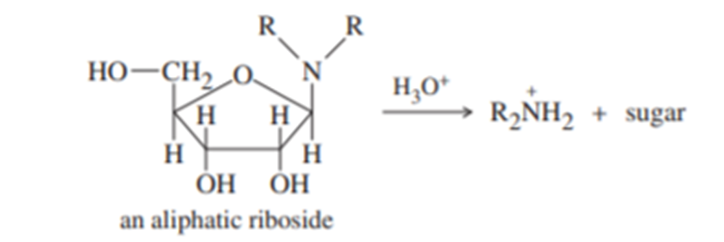
(b) Ribonucleosides are not so easily hydrolyzed, requiring relatively strong acid. Using your mechanism for part (a), show why cytidine and adenosine (for example) are not so readily hydrolyzed. Explain why this stability is important for living organisms.
Draw the following monosaccharides, using chair conformations for the pyranoses and Haworth projections for the furanoses.
H. G. Khorana won the Nobel Prize in Medicine in 1968 for developing the synthesis of DNA and RNA and for helping to unravel the genetic code. Part of the chemistry he developed was the use of selective protecting groups for the 5′ OH group of nucleosides.
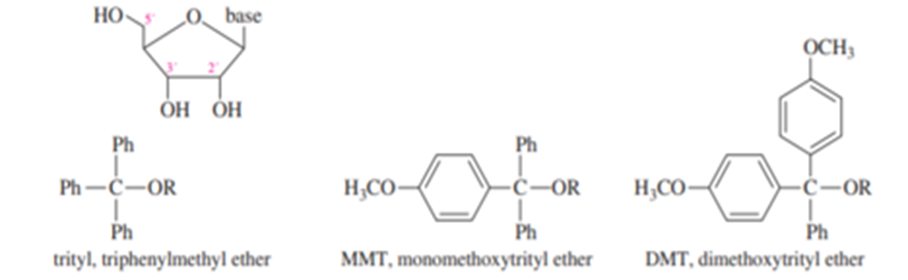
The trityl ether derivative of just the 5′ OH group is obtained by reaction of the nucleoside with trityl chloride, MMT chloride, or DMT chloride and a base like Et3N. The trityl ether derivative can be removed in dilute aqueous acid. DMT derivatives hydrolyze fastest, followed by MMT derivatives, and trityl derivatives slowest.
(a) Draw the product with the trityl derivative on the 5′ oxygen.
(b) Explain why the trityl derivative is selective for the 5′ OH group. Why doesn’t it react at 2′ or 3′? (c) Why is the DMT group easiest to remove under dilute acid conditions? Why does the solution instantly turn orange when acid is added to a DMT derivative?
An important protecting group developed specifically for polyhydroxy compounds like nucleosides is the tetraisopropyl-disiloxanyl group, abbreviated TIPDS, that can protect two alcohol groups in a molecule.
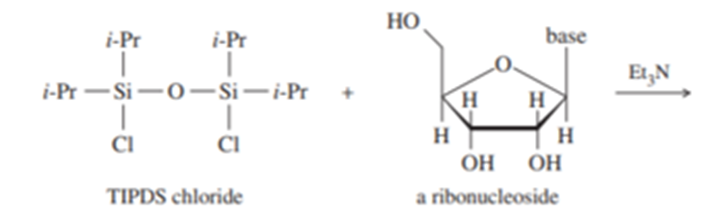
(a) The TIPDS group is somewhat hindered around the Siatoms by the isopropyl groups. Which OHis more likely to react first with TIPDS chloride? Show the product with the TIPDS group on one oxygen.
(b) Once the TIPDS group is attached at the first oxygen, it reaches around to the next closest oxygen. Show the final product with two oxygens protected.
(c) The unprotected hydroxy group can now undergo reactions without affecting the protected oxygens. Show the product after the protected nucleoside from (b) is treated with tosyl chloride and pyridine, followed by NaBr, ending with deprotection with Bu4NF.
Question. Retroviruses like HIV, the pathogen responsible for AIDS, incorporate an RNA template that is copied into DNA during infection. The reverse transcriptaseenzyme that copies RNA into DNA is relatively nonselective and error-prone, leading to a high mutation rate. Its lack of selectivity is exploited by the anti-HIV drug AZT (3’-azido-2’,3’-dideoxythymidine), which becomes phosphorylated and is incorporated by reverse transcriptase into DNA, where it acts as a chain terminator. Mammalian DNA polymerases are more selective, having a low affinity for AZT, so its toxicity is relatively low.
What do you think about this solution?
We value your feedback to improve our textbook solutions.