The relative configurations of the stereoisomers of tartaric acid were established by the following synthesis:
(1) D-(+)-glyceraldehyde diastereomers A and B (separated)
diastereomers A and B (separated)
(2) Hydrolysis of A and B using aqueous Ba(OH)2 gave C and D, respectively.
(3) HNO3 oxidation of C and D gave (-)-tartaric acid and meso-tartaric acid, respectively.
(a) You know the absolute configuration of D-(+)-glyceraldehyde, Use Fischer projections to show the absolute configurations of products A, B, C, and D.
(b) Show the absolute configurations of the three stereoisomers of tartaric acid: (+)-tartaric acid, (-)-tartaric acid, and meso-tartaric acid.
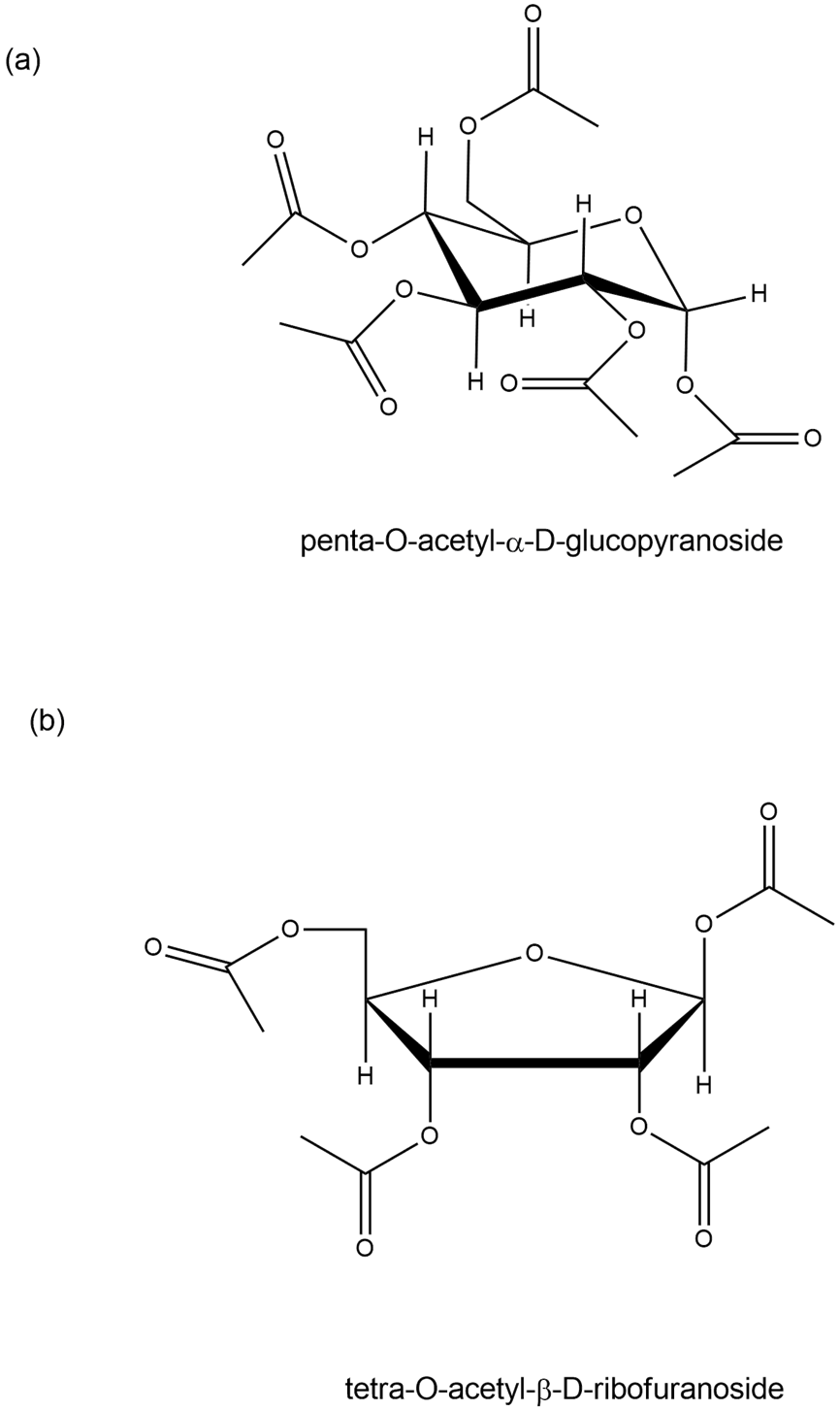






 diastereomers A and B (separated)
diastereomers A and B (separated)