Chapter 23: Q6P. (page 1214)
Question:
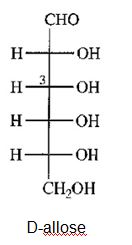
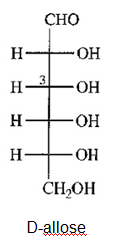
a) Draw D-allose, the C3 epimer of glucose.
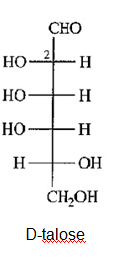
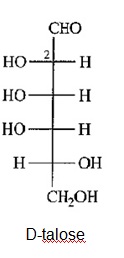
b) Draw D-talose, the C2 epimer of D-galactose.
c) Draw D-idose, the C3 epimer of D-talose. Now compare your answers with Figure 23-3.
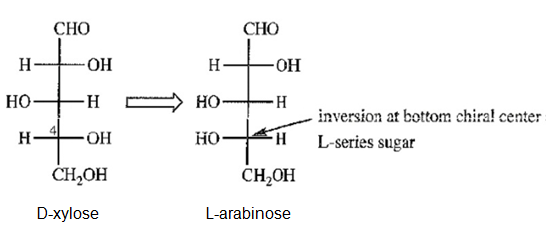
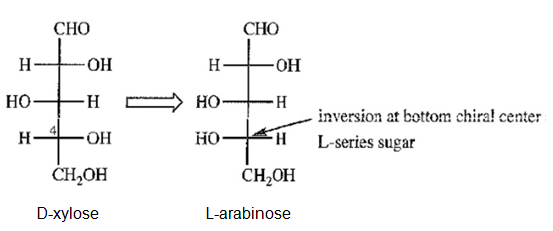
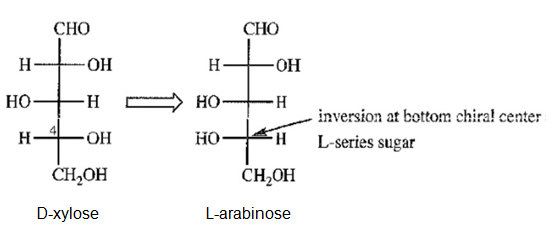
d) Draw the C4 “epimer” of D-xylose. Notice that this “epimer” is actually an L-series sugar, and we have seen its enantiomer. Give the correct name for this L-series sugar.
Short Answer
Answer
(a)
(b)
(c)
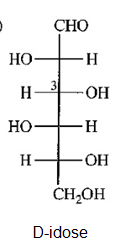
(d)