Chapter 23: Q24P (page 1226)
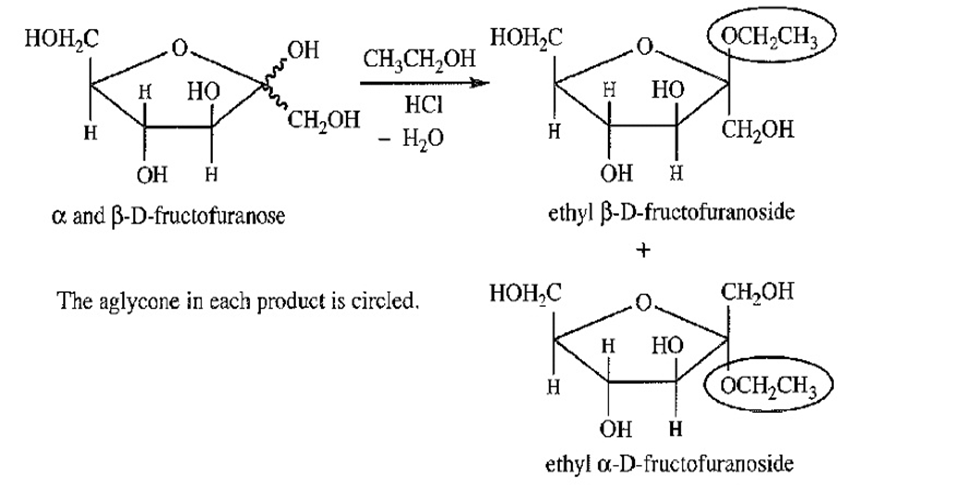
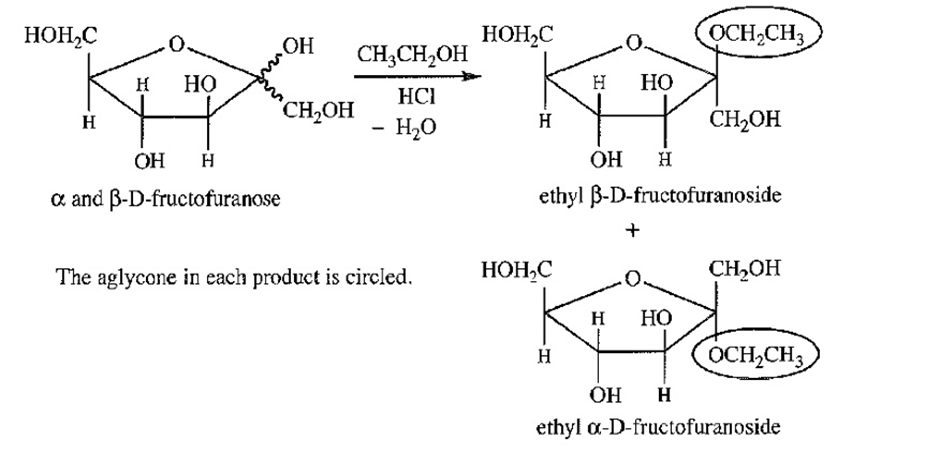
Treatment of either anomer of fructose with excess ethanol in the presence of a trace of HCI gives a mixture of the α and β anomers of ethyl-D-fructofuranoside. Draw the starting materials, reagents, and products for this reaction. Circle the aglycone in each product.
Short Answer