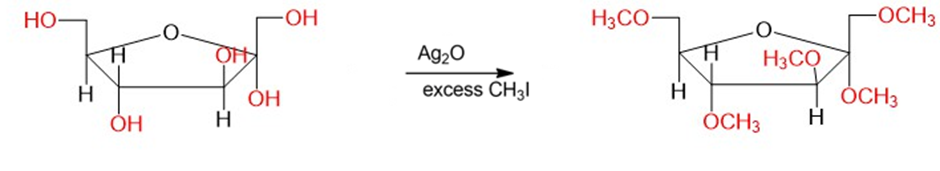
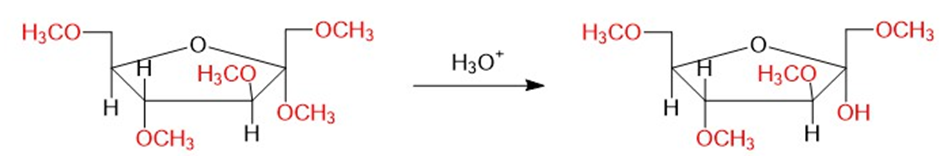
Chapter 23: Q25P (page 1228)
(a) Show the product that results when fructose is treated with an excess of methyl iodide and silver oxide.
(b) Show what happens when the product of part (a) is hydrolyzed using dilute acid.
(c) What do the results of parts (a) and (b) imply about the hemiacetal structure of fructose?
Short Answer
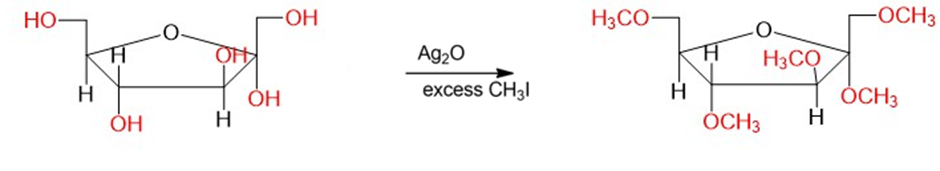
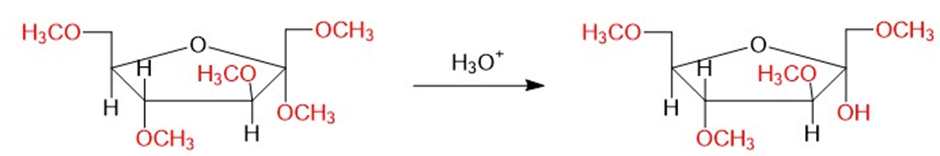
- The results show that the acetals are easily hydrolyzed by dilute acid whereas ethers are stable under these conditions.