Chapter 23: Q26P (page 1228)
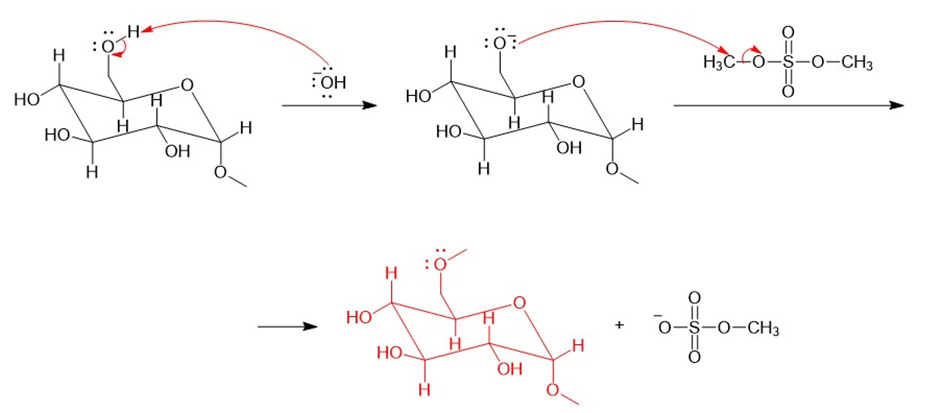
Question: Propose a mechanism for methylation of any one of the hydroxy groups of ,using and dimethyl sulfate.
Short Answer
Learning Materials
Features
Discover
Chapter 23: Q26P (page 1228)
Question: Propose a mechanism for methylation of any one of the hydroxy groups of ,using and dimethyl sulfate.

All the tools & learning materials you need for study success - in one app.
Get started for free
(a) Figure 23-2 shows that the degradation of D-glucose gives D-arabinose, an aldopentose. Arabinose is most stable in its furanose form. Draw D-arabinofuranose.
(b) Ribose, the C2 epimer of arabinose, is most stable in its furanose form. Draw D-ribofuranose.
Which of the following sugars are reducing sugars? Comment on the common name sucrose for table sugar.
(a) (b) (an aldohexose)
(c) (d)
(e) (f)

Cellulose is converted to cellulose acetateby treatment with acetic anhydride and pyridine. Cellulose acetate is soluble in common organic solvents, and it is easily dissolved and spun into fibers. Show the structure of cellulose acetate.
Raffinose is a trisaccharide (C18H32O16) isolated from cottonseed meal. Raffinose does not reduce Tollens reagent, and it does not mutarotate. Complete hydrolysis of raffinose gives D-glucose, D-fructose, and D-galactose. When raffinose is treated with invertase, the products are D-fructose and a reducing disaccharide called melibiose. Raffinose is unaffected by treatment with a-galactosidase, but an α -galactosidase hydrolyzes it to D-galactose and sucrose. When raffinose is treated with dimethyl sulfate and base followed by hydrolysis, the products are 2,3,4-tri-O-methylglucose, 1,3,4,6-tetra-O-methylfructose, and 2,3,4,6-tetra-O-methylgalactose. Determine the complete structures of raffinose and melibiose and give a systematic name for melibiose.
Cytosine, uracil and guanine have tautomeric forms with aromatic hydroxyl groups. Draw these tautomeric forms.
What do you think about this solution?
We value your feedback to improve our textbook solutions.