Chapter 23: Q40P (page 1238)
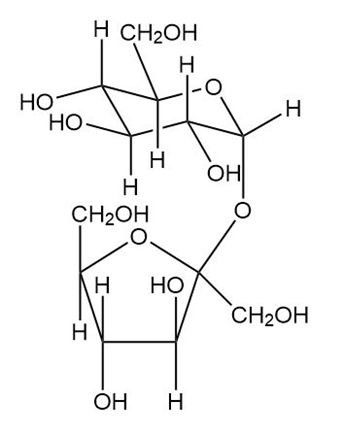
Trehalose is a nonreducing disaccharide (C12H22O11) isolated from the poisonous mushroom Amanita muscaria. Treatment with an α -glucosidase converts trehalose to two molecules of glucose, but no reaction occurs when trehalose is treated with a β -glucosidase. When trehalose is methylated by dimethyl sulfate in mild base and then hydrolyzed, the only product is 2,3,4,6-tetra-O-methylglucose. Propose a complete structure and systematic name for trehalose.
Short Answer
Trehalose:
α -D-glucopyranosyl-α -D-glucopyranoside: