Chapter 23: PROBLEM 23.66 (page 1257)
Question: Exposure to nitrous acid (see Section 19-16), sometimes found in cells, can convert cytosine to uracil.
- Propose a mechanism for this conversion.
- Explain how this conversion would be mutagenic upon replication.
- DNA generally includes thymine, rather than uracil(found in RNA). Based on this fact, explain why the nitrous acid-induced mutation of cytosine to uracil is more easily repaired in DNA than it is in RNA.
Short Answer
Answer
a. The mechanism for the conversion is:
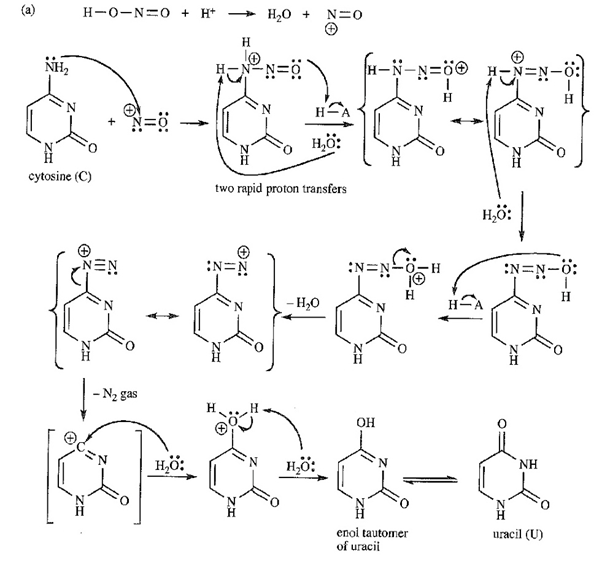
b . Conversion of cytosine to uracil will be mutagenic when a wrong base is inserted in a nucleic acid chain.
c. If cytosine gets diazotized to uracil, the DNA repair enzymes may detect it as a mutation and correct it. In DNA, thymine is used instead of uracil.
 The mechanism for the conversion
The mechanism for the conversion

 diastereomers A and B (separated)
diastereomers A and B (separated)