Chapter 23: Q34P (page 1232)
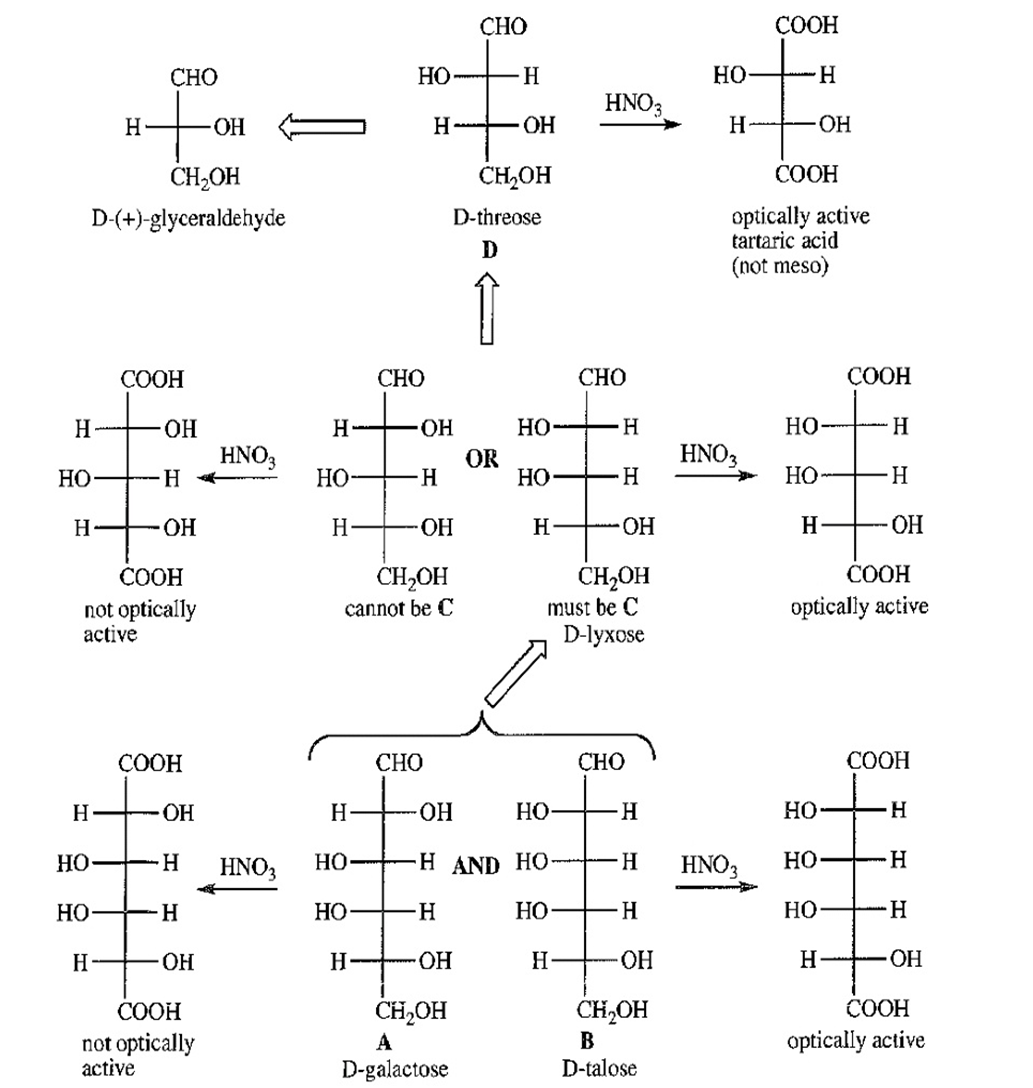
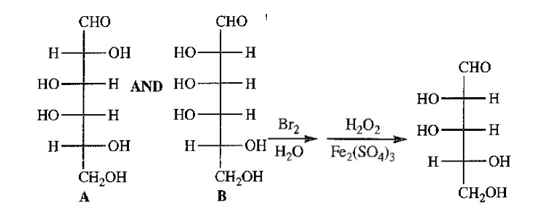
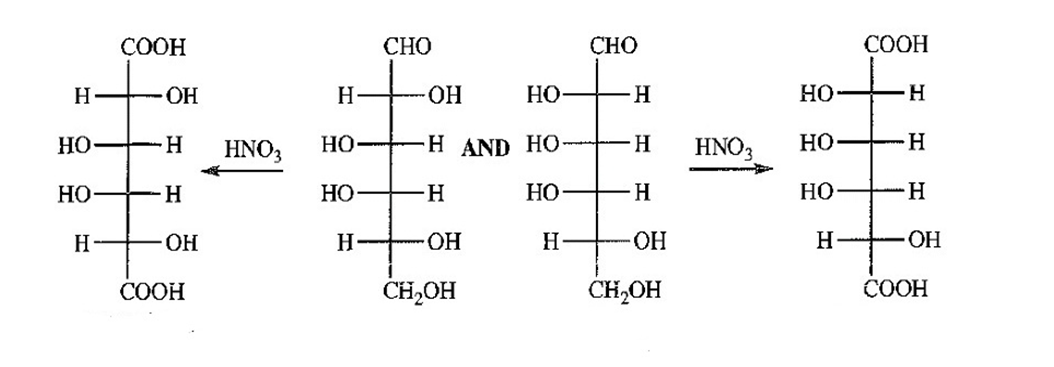
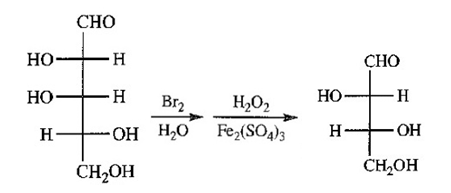
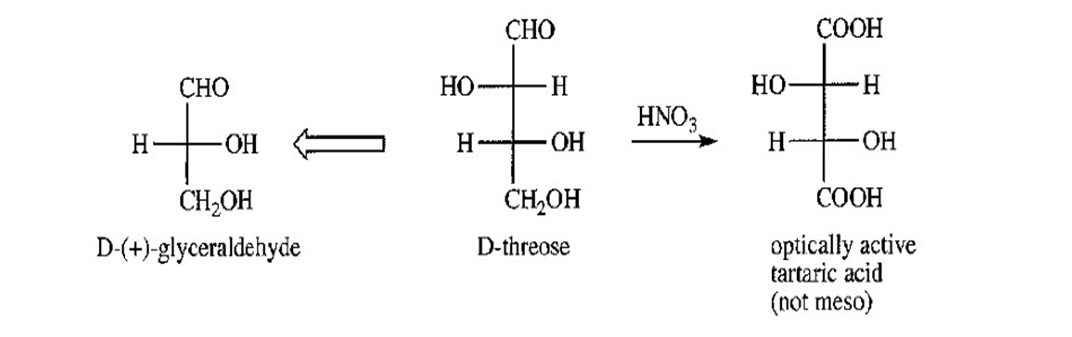
Aldohexoses A and B both undergo Ruff degradation to give aldopentose C. On treatment with warm nitric acid, aldopentose C gives an optically active aldaric acid. B also reacts with warm nitric acid to give an optically active aldaric acid, but A reacts to give an optically inactive aldaric acid. Aldopentose C is degraded to aldotetrose D, which gives optically active tartaric acid when it is treated with nitric acid. Aldotetrose D is degraded to (+)-glyceraldehyde. Deduce the structures of sugars A,B,C and D, and use Figure 23-3 to determine the correct names of these sugars.
Short Answer